HistoSonics Moves to Advance Additional Histotripsy Applications Announcing FDA Submission for Kidney Tumors
11.5.2026 14:00:00 CEST | Business Wire | Press release
HistoSonics, the developer of the Edison® Histotripsy System and novel histotripsy therapy platform, today announced it has submitted a De Novo request to the U.S. Food and Drug Administration seeking authorization to expand the use of its Edison® Histotripsy System to include the destruction of kidney (renal) tumors.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260511268688/en/
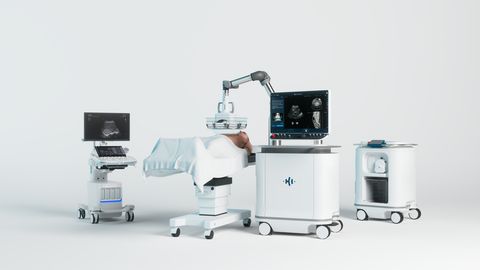
HistoSonics Edison® Histotripsy System
This milestone marks a significant step forward in the company’s mission to transform the treatment of solid tumors with a completely non-invasive technology that harnesses focused ultrasound to mechanically liquefy and destroy targeted tissue, reducing the risk of many complications and side effects associated with surgery, radiation, and other common therapies.
“This submission is an important milestone in expanding histotripsy beyond the liver and into the kidney, an area where patients and physicians are seeking organ-preserving and nephron-sparing alternatives,” said Mike Blue, Chairman and CEO of HistoSonics. “We believe histotripsy has the potential to fundamentally change how solid tumors are treated, delivering precise, non-invasive therapy that preserves what matters most – including patient quality of life.”
The submission is supported by clinical evidence from the ongoing #HOPE4KIDNEY Trial (NCT05820087), a prospective, multi-center, single-arm pivotal trial evaluating the safety and effectiveness of the Edison® System in destroying targeted kidney tumors. The trial enrolled 67 patients and continues to generate promising insights into the potential of histotripsy in kidney applications.
“Histotripsy represents a fundamentally novel approach to treating kidney tumors. In the #HOPE4KIDNEY Trial, we observed precise, noninvasive tumor destruction without incisions, radiation, or thermal injury. These findings highlight the potential of histotripsy to expand treatment options for patients with kidney tumors and represent an important step forward for both physicians and patients,” said Dr. William Huang, Urologic Oncologist at NYU Langone Health and Principal Investigator for the #HOPE4KIDNEY Trial.
Kidney tumors represent a significant and growing clinical challenge, with approximately 680,000 people living with kidney tumors in the United States and an estimated 80,000 new cases will be diagnosed in 2026. Current standard-of-care treatments—such as partial nephrectomy and thermal ablation—are invasive and may carry risks including bleeding, infection, and unintended damage to surrounding tissue. Histotripsy offers a compelling new alternative: a non-invasive, non-thermal approach designed to precisely target tumors while helping to preserve critical kidney function.
Currently, the Edison System is indicated for the non-invasive destruction of liver tumors, including unresectable liver tumors, using histotripsy, a non-thermal, mechanical process of focused ultrasound. The system is currently being evaluated by the FDA for the destruction of kidney tumors, and its use in kidney applications remains investigational.
About HistoSonics
HistoSonics is a privately held medical device company developing a non-invasive platform and proprietary sonic beam therapy utilizing the science of histotripsy, a novel mechanism of action that uses focused ultrasound to mechanically destroy and liquefy unwanted tissue and tumors. The company is currently focused on commercializing their Edison System in the US and select global markets for liver treatment while expanding histotripsy applications into other organs like kidney, pancreas, prostate, and others. HistoSonics has offices in Ann Arbor, MI, Minneapolis, MN and Madison, WI. For more information on the Edison Histotripsy System please visit: www.histosonics.com. For patient-related information please visit: www.myhistotripsy.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260511268688/en/

Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
Only 7% of Companies Achieve Full Compliance as Global Expansion Increases Legal Complexity11.5.2026 15:00:00 CEST | Press release
47% of general counsels say beneficial ownership rules pose the biggest risks to legal operations44% lack confidence in meeting cross-border data security requirements As businesses accelerate their global expansion in 2026, compliance fails to keep pace. In fact, only 7% of organizations report full compliance across their global entities, according to a new study by CSC, the leading provider of global business administration and compliance solutions. CSC surveyed 350 general counsel (GCs) and senior legal professionals across Europe, North America, and Asia Pacific to examine how their teams navigate international expansion, regulatory pressure, and the increasing adoption of artificial intelligence (AI).¹ The findings appear in CSC’s latest report, General Counsel Barometer 2026: From Complexity to Control. Most organizations report partial compliance, with over half (53%) estimating they are 50–75% compliant, and a further 35% placing themselves between 76%–99%. This leaves just 7%
IFF Opens Vanilla Innovation Center in Madagascar11.5.2026 14:15:00 CEST | Press release
Advancing science‑led flavor innovation where vanilla is grown IFF (NYSE: IFF)—a global leader in flavors, fragrances, food ingredients, health & bioscience—today announced the opening of its Vanilla Innovation Center in Madagascar, reinforcing vanilla as a strategic and priority tonality for IFF and strengthening its ability to innovate at origin. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260508110162/en/ IFF's Vanilla Innovation Center in Madagascar “The opening of the center marks an important step in how we approach vanilla innovation,” said Adam Jańczuk, Ph.D., senior vice president, research, creation and design, Taste, IFF. “By strengthening our presence at origin, we connect science, creativity and sustainability more closely, responding to climate changes, safeguarding quality and creating value across the supply chain.” Located in Toamasina, Madagascar’s principal seaport, near vanilla growing areas and post‑h
ARIS Recognised as a Leader in Gartner® Magic Quadrant™ for Process Intelligence Platforms, Believes This Reinforces Its Role in Enabling Enterprise AI at Scale11.5.2026 14:00:00 CEST | Press release
ARIS, the process context foundation platform for enterprise AI deployment, today announced its recognition as a Leader in the Gartner® Magic Quadrant™ for Process Intelligence Platforms. This is the fourth consecutive year that ARIS has been recognized as a Leader in the report and the company believes it underscores a continued commitment to innovation and growth as enterprises focus on turning AI ambition into measurable business outcomes. While technology has advanced rapidly, companies are struggling to operationalise AI across complex operating models. ARIS sees this recognition by Gartner as a reflection of its strength in delivering a single unified platform for process intelligence, providing the context layer on which G2000 organisations can successfully deploy and scale agentic AI. “AI is moving from experimentation to execution – but many enterprises are finding it difficult to scale,” said Guillaume Bacuvier, CEO of ARIS. “The reason is simple: AI lacks the context it need
Logistics Reply Named a Visionary in 2026 Gartner® Magic Quadrant™ for Warehouse Management Systems and Ranks #2 for Level 2 and #3 for Level 3 Operations Use Cases in Gartner® Critical Capabilities Report11.5.2026 14:00:00 CEST | Press release
Logistics Reply, the Reply Group company specializing in innovative solutions for supply chain execution, is proud to announce its recognition as a Visionary in the Gartner® Magic Quadrant™ for Warehouse Management Systems for the seventh consecutive year, as its global team of warehouse technology professionals continues to drive innovation that puts customers first. Additionally, Logistics Reply for its LEA Reply™ WMS is recognized in the 2026 Gartner® Critical Capabilities for Warehouse Management Systems report where it ranked #2 for Level 2 Warehouse Operations and #3 for Level 3 Warehouse Operations. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260511344452/en/ We believe these important recognitions underscore Logistics Reply's commitment to delivering intelligent, flexible and scalable warehouse execution solutions for enterprise customers around the world. For us, our placement in the Visionaries Quadrant reflects
L&T Technology Services, Assai Software Partner to Advance Digital Twin-led Engineering Intelligence11.5.2026 12:30:00 CEST | Press release
Partnership in LTTS’ Sustainability segment strengthens AI-powered solutions for global energy leaders L&T Technology Services (BSE: 540115, NSE: LTTS), a global leader in AI, Digital & ER&D Consulting Services signed a strategic agreement with Europe-based Assai Software Services, a leading Engineering Document Management Systems (EDMS) and Digital Twin solutions provider to develop advanced technology solutions and cater to the massive energy & utilities sector in Europe. The partnership in LTTS’ Sustainability segment will combine its engineering and delivery capabilities with Assai’s software expertise to address the growing complexity of asset-intensive projects for global energy leaders. By enabling seamless integration with Digital Twin technology, the alliance will deliver real-time asset visualization, traceability and governed engineering information across project lifecycles. LTTS has also established a dedicated Centre of Excellence (CoE) leveraging Assai’s platform to supp
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom