Printing the Future of Oncology: CTIBIOTECH Invests €3M and Secures €1.25M from France 2030 to Revolutionize Personalized Cancer Care
28.4.2026 14:28:00 CEST | Business Wire | Press release
CTIBIOTECH™, a leading innovator in advanced human tissue engineering, proudly announces CTIONCOTEST™ platform is awarded Major Bpifrance Funding to Industrialize 3D Bioprinted Cancer Models
CTIBIOTECH Announces the CTIONCOTEST ™ Project to Revolutionize Cancer Research with 3D Bioprinted Microtumors, Backed by France 2030 and Bpifrance
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428242352/en/

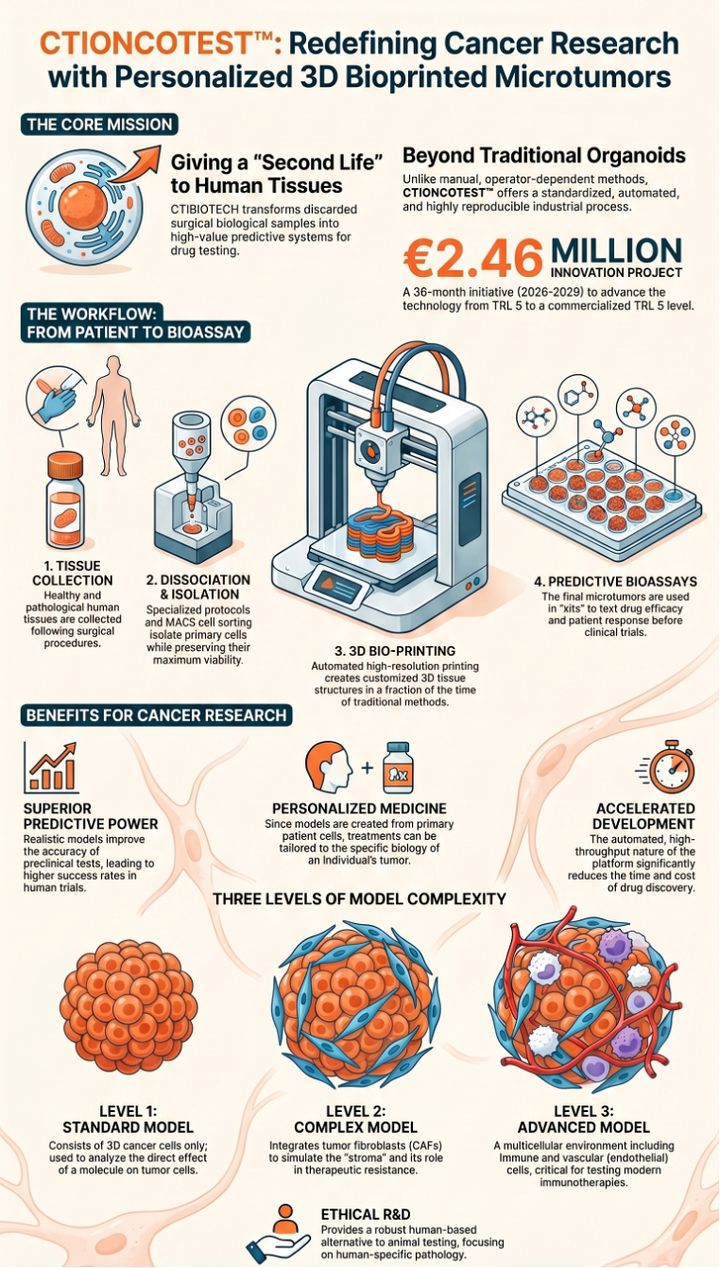
The CTIONCOTEST™ platform by CTIBIOTECH is an innovative 3D bioprinting technology that revolutionizes preclinical cancer research . It provides an automated, industrial-scale process to produce high-throughput, patient-specific human "microtumors" directly from primary tumor cells . Designed to tackle the 95% clinical failure rate of new cancer drugs, it faithfully replicates the complex tumor microenvironment, including critical hypoxic and necrotic zones . Targeting high-mortality breast, colon, and pancreatic cancers, the platform features three complexity levels: standard (cancer cells), intermediate (+fibroblasts), and advanced (+immune/endothelial cells) . A key breakthrough is its record cellular viability of 128 days, enabling unprecedented long-term studies on drug resistance and metastasis . Ultimately, CTIONCOTEST delivers a highly predictive, 100% human-derived ethical alternative to animal testing.
CTIBIOTECH™, an innovative French Contract Research, Development, and Manufacturing Organization (CRDMO) based in Meyzieu-Lyon, proudly announces the launch of the CTIONCOTEST ™ project, a groundbreaking €3 million initiative aimed at transforming preclinical cancer research. To accelerate this critical innovation, CTIBIOTECH ™ has been awarded €1.25 million in funding from Bpifrance under the "Aide au Développement Deeptech" program, with the strategic support of France 2030 and the Government of France.
Currently, 95% of new cancer drugs fail during human clinical trials, costing the pharmaceutical industry billions of euros and delaying life-saving treatments for patients. The CTIONCOTEST ™ project addresses this urgent global health challenge by developing an automated, industrial-scale process to 3D bioprint hundreds of standardized human "microtumors" directly from a specific cancer patient's primary tumor cells.
Over a 36-month period, the project will advance the technology from a working prototype (TRL 5) to a fully functional, high-throughput commercial product (TRL 8) ready for market entry by 2029.
Key highlights of the CTIONCOTEST ™ project include:
- CTIONCOTEST ™ is a de-risking tool for the pharmaceutical industry, where 95% of new treatments currently fail in clinical trials due to poor preclinical predictivity.
- Market Drivers: Rising global cancer mortality (projected 12M deaths by 2030) and regulatory pressure to adopt New Approach Methodologies (NAMs) to replace animal testing.
- Business Model: Dual-revenue strategy involving the sale of turnkey kits and contract research/innovation (CRDMO) for biopharma partners.
- Future Vision: Establishing global leadership in custom bioproduction for immuno-oncology and personalized medicine
- Focus on High-Mortality Cancers: The project will specifically develop functional 3D models for breast, colon, and pancreatic cancers—areas that currently suffer from a severe lack of reliable experimental models.
- Unprecedented Biological Fidelity: Unlike manual methods, CTIBIOTECH’s computer-aided 3D bioprinting perfectly mimics the complex tumor microenvironment, including hypoxic and necrotic zones. These models have demonstrated a record-breaking cellular viability of 128 days, allowing researchers to conduct unprecedented long-term studies on drug resistance and cancer metastasis.
- Ethical Alternatives to Animal Testing: By providing a highly predictive, 100% human-derived testing platform, CTIONCOTEST ™ actively supports the regulatory shift towards New Approach Methodologies (NAMs), drastically reducing and replacing the need for animal experimentation in preclinical trials.
- A Collaborative Ecosystem of Excellence: Labilized by the Lyon biopôle Auvergne Rhone-Alpes competitiveness cluster, the project integrates a world-class network of academic, clinical, and industrial partners. Key collaborators include the Centre Léon Bérard, Hospices Civils de Lyon (HCL), Medical University Plovdiv, Transgene, GenXMap, Oncodesign Services, and Novotec.
Professor Colin McGuckin, President and Chief Scientific Officer of CTIBIOTECH shared his pride and vision "The industrialization of the CTIONCOTEST platform marks a crucial step toward personalized medicine against cancer, where treatments can be individually tailored to a patient's specific tumor profile".
"This financial support from the French State represents a vital lever to maximize the impact of our technology, allowing us to advance twice as fast in developing and commercializing these 3D bioprinted microtumors for transform cancer drug development" added Dr Nico Forraz, Chief Executive Officer of CTIBIOTECH
The CTIONCOTEST™ project will directly contribute to local economic growth, creating 5 immediate permanent scientific positions in 2026, with projections to create 51 jobs and 16 million euros revenue by 2031. Research, development, and industrialization efforts will be conducted at CTIBIOTECH’s 800m² state-of-the-art facility and certified biobank in Meyzieu, Metropolis of Lyon, France.
About CTIBIOTECH™: CTIBIOTECH™ is a global biotechnology company specializing leader in 3D bioprinting and advanced human tissue engineering. By transforming discarded human surgical tissues into high-value predictive bioassays, CTIBIOTECH™ provides integrated drug discovery, development and innovation solutions for the pharmaceutical, medical device, and cell therapy industries globally.
Note to Editors: High-resolution images and interviews with CTIBIOTECH™ executives are available upon request.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260428242352/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
HTEC Research: Only One in Three Healthcare Organizations is Ready to Scale AI28.4.2026 15:22:00 CEST | Press release
Healthcare and life sciences leaders are advancing AI with caution—fragmentation, capability gaps, and execution challenges are slowing enterprise-wide impact AI is already embedded across healthcare and life sciences. Most organizations are deploying it, and confidence in its potential is high. Yet for many, the real challenge is only just beginning. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428872907/en/ HTEC, a global AI‑first provider of software and hardware design and engineering services, today released new research based on a global survey of 253 C-level HLS executives across the United States, United Kingdom, Germany, Spain, Saudi Arabia, and the UAE. AI is already embedded across healthcare and life sciences. Most organizations are deploying AI, and confidence in its potential is high. Yet for many, the real challenge is only just beginning. HTEC, a global AI‑first provider of software and hardware design a
JPMorganChase Named First-Ever Global Banking Partner of the Olympic Games28.4.2026 15:00:00 CEST | Press release
International Olympic Committee and JPMorganChase Announce Landmark Global Olympic Partnership The International Olympic Committee (IOC) and JPMorganChase today announced a landmark Worldwide Olympic Partnership, making JPMorganChase the first Global Banking Partner in Olympic history. The partnership includes the Los Angeles 2028 Olympic and Paralympic Games (LA28 Games) and the French Alps 2030 Olympic and Paralympic Winter Games. The firm has also reached an agreement with LA28 to become the Official Bank of Team USA and LA28, and a Founding Partner of the LA28 Games. The partnership reflects a shared commitment to ambition and excellence, and places athletes and communities at its core. Kirsty Coventry, President of the IOC, commented: “JPMorganChase is the first Global Partner from the banking sector in Olympic history, and we are proud to welcome them to the Worldwide Olympic Partner programme. This partnership reflects our shared values of ambition, excellence and will support t
BlueSnap, Powered by Payroc, Launches Local Acquiring in New Zealand to Boost Approval Rates and Reduce Cross-Border Costs28.4.2026 15:00:00 CEST | Press release
BlueSnap, powered by Payroc, today announced the availability of local acquiring in New Zealand, expanding its global payment orchestration capabilities and enabling businesses to process transactions domestically. Local acquiring allows businesses with a legal entity in New Zealand to process payments through domestic acquiring banks rather than routing transactions cross-border. This helps increase authorization rates, reduce payment failures, and lower transaction costs, critical factors for businesses selling into the region. Why Local Acquiring in New Zealand Matters For B2B and B2C businesses operating globally, cross-border payments often lead to higher fees and lower approval rates. Building local acquiring infrastructure independently requires managing multiple bank relationships, compliance, and integrations, making it costly and complex. BlueSnap simplifies this by providing local acquiring through a single platform, allowing businesses with a legal entity in New Zealand to
Vensica Medical Receives FDA IND Clearance to Initiate Phase 2 Study of ViXe, Its Needle-Free Xeomin® Delivery System for Overactive Bladder28.4.2026 14:42:00 CEST | Press release
Vensica Medical, a clinical-stage company developing needle-free therapeutic delivery solutions for urologic diseases, today announced that the U.S. Food and Drug Administration (FDA) has cleared the Company’s Investigational New Drug (IND) application to initiate a Phase 2 clinical trial of ViXe. The study will evaluate the Vibe® ultrasound-based, needle-free drug delivery system in combination with Xeomin® (incobotulinumtoxinA) for the treatment of overactive bladder (OAB). This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260414145279/en/ Avner Geva, Cofounder, CEO (left), and Avi Eftel, Cofounder, CTO (right) Overactive bladder affects an estimated 33 million adults in the United States and is characterized by urinary urgency, frequency, and incontinence. While intravesical botulinum toxin injection is an established second-line treatment, the invasive procedure limits patient access and acceptance. The ViXe program is desi
BMG and Concord Combine to Create World’s Leading Independent Music Company28.4.2026 14:11:00 CEST | Press release
Creates global platform to deliver superior service to artists, songwriters, and partnersUnlocks meaningful value through greater scale and strong cash flow, enabling continued investment in music rights, creative talent, and technologyBob Valentine to serve as CEO and Thomas Coesfeld as Chairman of the combined company, with global headquarters in Nashville and European Headquarters in BerlinCombined company to be named BMG with divisions “BMG Publishing” and “Concord Records” BMG and Concord today announced a definitive agreement to combine their businesses, creating the leading independent music company in the world. Operating under the BMG name, the combined company will bring together two highly complementary businesses to establish a scaled, global company delivering superior service to artists, songwriters, and partners – firmly grounded in an independent, artist-first model. This press release features multimedia. View the full release here: https://www.businesswire.com/news/ho
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom