Printing the Future of Oncology: CTIBIOTECH Invests €3M and Secures €1.25M from France 2030 to Revolutionize Personalized Cancer Care
28.4.2026 14:28:00 CEST | Business Wire | Press release
CTIBIOTECH™, a leading innovator in advanced human tissue engineering, proudly announces CTIONCOTEST™ platform is awarded Major Bpifrance Funding to Industrialize 3D Bioprinted Cancer Models
CTIBIOTECH Announces the CTIONCOTEST ™ Project to Revolutionize Cancer Research with 3D Bioprinted Microtumors, Backed by France 2030 and Bpifrance
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428242352/en/

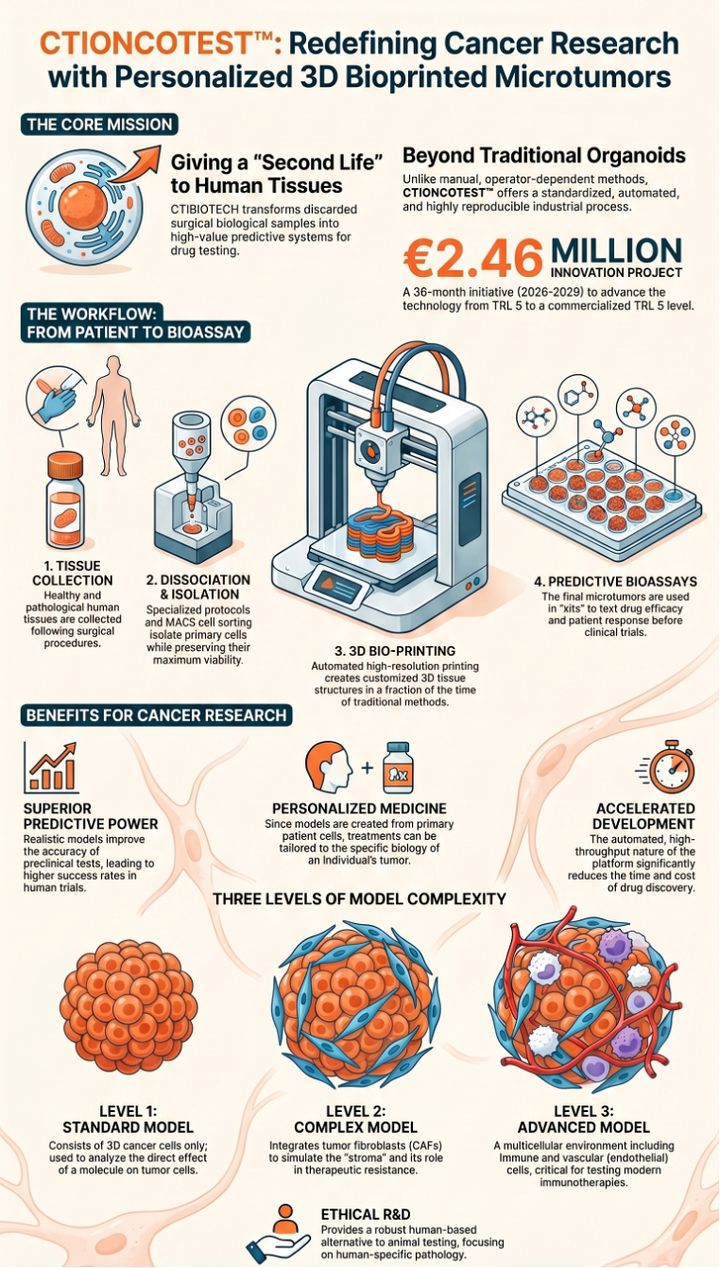
The CTIONCOTEST™ platform by CTIBIOTECH is an innovative 3D bioprinting technology that revolutionizes preclinical cancer research . It provides an automated, industrial-scale process to produce high-throughput, patient-specific human "microtumors" directly from primary tumor cells . Designed to tackle the 95% clinical failure rate of new cancer drugs, it faithfully replicates the complex tumor microenvironment, including critical hypoxic and necrotic zones . Targeting high-mortality breast, colon, and pancreatic cancers, the platform features three complexity levels: standard (cancer cells), intermediate (+fibroblasts), and advanced (+immune/endothelial cells) . A key breakthrough is its record cellular viability of 128 days, enabling unprecedented long-term studies on drug resistance and metastasis . Ultimately, CTIONCOTEST delivers a highly predictive, 100% human-derived ethical alternative to animal testing.
CTIBIOTECH™, an innovative French Contract Research, Development, and Manufacturing Organization (CRDMO) based in Meyzieu-Lyon, proudly announces the launch of the CTIONCOTEST ™ project, a groundbreaking €3 million initiative aimed at transforming preclinical cancer research. To accelerate this critical innovation, CTIBIOTECH ™ has been awarded €1.25 million in funding from Bpifrance under the "Aide au Développement Deeptech" program, with the strategic support of France 2030 and the Government of France.
Currently, 95% of new cancer drugs fail during human clinical trials, costing the pharmaceutical industry billions of euros and delaying life-saving treatments for patients. The CTIONCOTEST ™ project addresses this urgent global health challenge by developing an automated, industrial-scale process to 3D bioprint hundreds of standardized human "microtumors" directly from a specific cancer patient's primary tumor cells.
Over a 36-month period, the project will advance the technology from a working prototype (TRL 5) to a fully functional, high-throughput commercial product (TRL 8) ready for market entry by 2029.
Key highlights of the CTIONCOTEST ™ project include:
- CTIONCOTEST ™ is a de-risking tool for the pharmaceutical industry, where 95% of new treatments currently fail in clinical trials due to poor preclinical predictivity.
- Market Drivers: Rising global cancer mortality (projected 12M deaths by 2030) and regulatory pressure to adopt New Approach Methodologies (NAMs) to replace animal testing.
- Business Model: Dual-revenue strategy involving the sale of turnkey kits and contract research/innovation (CRDMO) for biopharma partners.
- Future Vision: Establishing global leadership in custom bioproduction for immuno-oncology and personalized medicine
- Focus on High-Mortality Cancers: The project will specifically develop functional 3D models for breast, colon, and pancreatic cancers—areas that currently suffer from a severe lack of reliable experimental models.
- Unprecedented Biological Fidelity: Unlike manual methods, CTIBIOTECH’s computer-aided 3D bioprinting perfectly mimics the complex tumor microenvironment, including hypoxic and necrotic zones. These models have demonstrated a record-breaking cellular viability of 128 days, allowing researchers to conduct unprecedented long-term studies on drug resistance and cancer metastasis.
- Ethical Alternatives to Animal Testing: By providing a highly predictive, 100% human-derived testing platform, CTIONCOTEST ™ actively supports the regulatory shift towards New Approach Methodologies (NAMs), drastically reducing and replacing the need for animal experimentation in preclinical trials.
- A Collaborative Ecosystem of Excellence: Labilized by the Lyon biopôle Auvergne Rhone-Alpes competitiveness cluster, the project integrates a world-class network of academic, clinical, and industrial partners. Key collaborators include the Centre Léon Bérard, Hospices Civils de Lyon (HCL), Medical University Plovdiv, Transgene, GenXMap, Oncodesign Services, and Novotec.
Professor Colin McGuckin, President and Chief Scientific Officer of CTIBIOTECH shared his pride and vision "The industrialization of the CTIONCOTEST platform marks a crucial step toward personalized medicine against cancer, where treatments can be individually tailored to a patient's specific tumor profile".
"This financial support from the French State represents a vital lever to maximize the impact of our technology, allowing us to advance twice as fast in developing and commercializing these 3D bioprinted microtumors for transform cancer drug development" added Dr Nico Forraz, Chief Executive Officer of CTIBIOTECH
The CTIONCOTEST™ project will directly contribute to local economic growth, creating 5 immediate permanent scientific positions in 2026, with projections to create 51 jobs and 16 million euros revenue by 2031. Research, development, and industrialization efforts will be conducted at CTIBIOTECH’s 800m² state-of-the-art facility and certified biobank in Meyzieu, Metropolis of Lyon, France.
About CTIBIOTECH™: CTIBIOTECH™ is a global biotechnology company specializing leader in 3D bioprinting and advanced human tissue engineering. By transforming discarded human surgical tissues into high-value predictive bioassays, CTIBIOTECH™ provides integrated drug discovery, development and innovation solutions for the pharmaceutical, medical device, and cell therapy industries globally.
Note to Editors: High-resolution images and interviews with CTIBIOTECH™ executives are available upon request.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260428242352/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
BTG Pactual Timberland Investment Group Raises US$1.24 Billion for Largest Reforestation Fund to Date[1]28.4.2026 17:00:00 CEST | Press release
Milestone positions Latin American reforestation strategy as a leading model for scalable, high-integrity conservation and restoration—combining sustainable commercial reforestation with large-scale ecosystem conservation and restoration to target economic returns, climate and biodiversity gains, and rural community benefits. Conservation International serves as Impact Adviser for social and environmental outcomes. BTG Pactual Timberland Investment Group (BTG Pactual TIG), one of the world’s largest timberland managers, today announced the close of fundraising for its Latin American Reforestation Strategy, reaching US$1.24 billion in commitments. This represents the largest reforestation fund closed to date,1 providing a model for large-scale reforestation and restoration that seeks to deliver durable climate impact, tangible benefits for rural communities, biodiversity outcomes, and sustainable commercial production. Global environmental organization Conservation International serves
Qatar Implements Relief Measures to Support Business Community28.4.2026 16:56:00 CEST | Press release
Qatar has activated a comprehensive package of support measures designed to maintain market stability and reinforce investor confidence amid evolving regional conditions. These measures, delivered through the nation’s foreign direct investment ecosystem, provide immediate financial, regulatory and operational relief to Qatar’s international business community. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428904909/en/ Infographic highlights key relief measures implemented in Qatar to support the business community (Infographic: AETOSWire) H.E. Sheikh Faisal bin Thani Al Thani, Minister of Commerce and Industry and Chairman of Invest Qatar, stated: “Qatar’s commitment to its business community is unwavering. As regional conditions continue to evolve, we remain fully focused on acting decisively to support companies operating in our market, safeguard business continuity and reinforce confidence. Our approach is grounded i
Dubai’s Medcare Hospital Becomes the World’s First to Treat an Adult Spinal Muscular Atrophy (SMA) Patient Outside the US28.4.2026 15:57:00 CEST | Press release
Medcare Royal Speciality Hospital (MRSH) in Dubai has become the world’s first hospital outside the USA, to offer a newly licensed intrathecal gene therapy - Itvisma, to adult patients with Spinal Muscular Atrophy (SMA). This one-time treatment was recently administered to a 22-year-old Egyptian patient, who was diagnosed at 18 months and confined to a wheelchair for most of his life. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428038416/en/ Dr. Vivek Mundada with Medcare multidisciplinary medical team (Photo: AETOSWire) Last year, Medcare also became the first healthcare provider globally to administer the newly licensed Itvisma to a four-year-old international SMA patient. SMA is a rare neuromuscular disease that leads to progressive muscle weakness and loss of mobility, impacting movement, breathing, and swallowing. Until now, advancements in gene therapy for SMA were primarily restricted to children under the age o
Perpetual Atomics and QSA Global Fuel the World’s First Americium-Powered Radioisotope Heater Unit Core28.4.2026 15:48:00 CEST | Press release
In a landmark achievement for international space exploration, Perpetual Atomics, QSA Global, Inc. and the University of Leicester have completed the first historical production of an Americium-fuelled Radioisotope Heater Unit (RHU) full scale core containing ceramic form americium-241 pellets in a metal containment structure. This breakthrough builds on the successful americium pelleting work in late 2025 and on two decades of experience in leading the development of americium space nuclear power systems by the Leicester team. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428027123/en/ QSA Global and Perpetual Atomics Team Born out of ENDURE, a European Space Agency (ESA) project, and part of a transatlantic collaboration, this achievement provides the steppingstones for an industrial pathway to deliver radioisotope power systems for spacecraft enabling these to survive and thrive in the most extreme environments in the
Alipay Launches AI Payment Processing Product to Help Businesses and OPCs Thrive in the Agentic Economy28.4.2026 15:47:00 CEST | Press release
New solution enables businesses to monetize services through AI agents Alipay today launched a new AI payment processing product that enables businesses, large and small, including One Person Companies (OPCs) in the Chinese mainland, to receive payments seamlessly when autonomous AI agents, including OpenClaw-type agents, purchase their services. This new product is being launched as AI agents are increasingly executing tasks on behalf of users, from booking travel and comparing prices, to allocating computing power and buying tokens. The product is available on Alipay’s website (https://aipay.alipay.com/). With Alipay’s AI payment processing product, small and medium-sized businesses do not need to build complex payment or settlement systems. By simply onboarding their monetizable services, businesses can reach more customers and receive payment each time an AI agent purchases their service. Bocha, an AI-powered search tool offering web search support for AI applications including Dee
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom