New Study Finds Masimo SedLine® Brain Function Monitoring Offers Significant Advantages During Pediatric Anesthesia
EEG-Guided Anesthesia Using Masimo Technology Significantly Reduced Pediatric Anesthesia Emergence Delirium, Time to Emergence and Discharge, and Cost of Care by Minimizing Sevoflurane Exposure
Masimo (NASDAQ: MASI) today announced the findings of a randomized clinical trial published in JAMA Pediatrics in which Dr. Yasuko Nagasaka and colleagues at Tokyo Women’s Medical University demonstrated the ability of electroencephalogram (EEG)-guided anesthesia, using Masimo SedLine® Brain Function Monitoring, to improve anesthesia administration in children undergoing surgery.1 The researchers found that the use of SedLine led to a significant reduction in the amount of a commonly used inhalation anesthetic (sevoflurane) needed to maintain anesthesia in pediatric patients, reducing their exposure to the drug. In turn, the patients experienced a significantly lower rate of pediatric anesthesia emergence delirium, or PAED, which commonly manifests in confusion, agitation, or hallucinations experienced during recovery. When compared with standard practice, children monitored with SedLine also regained consciousness faster and could be safely discharged to the post-anesthesia care unit, or PACU, sooner – time savings that, the researchers noted, may represent additional cost savings. In sum, the researchers found that Masimo SedLine helped to make pediatric anesthesia safer, more efficient, and more cost effective.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250610391732/en/
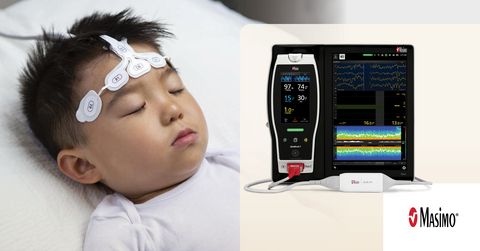
Masimo SedLine® Brain Function Monitoring
Children monitored with SedLine were exposed to an average of 1.4 MAC hours less sevoflurane and had a 14% lower incidence rate of PAED. On average, they regained consciousness 53% sooner and were discharged from the PACU 48% sooner. The time saved led to an estimated cost savings of $240 to $920 per patient.
PAED is a frequent and clinically significant complication in pediatric anesthesia,2 often leading to short-term distress among pediatric patients, parents, and staff. Some of the negative postoperative behaviors can persist for weeks or months.3 Exposure to commonly used volatile anesthetics like sevoflurane gas has been shown to contribute to PAED.4 In current standard practice, a fixed amount of sevoflurane, 1.0 minimum alveolar concentration (MAC), is used to maintain pediatric anesthesia, but as the authors note, that may be excessive.5 Dr. Nagasaka and her team hypothesized that by using bilateral EEG – in particular, the waveforms and multitaper density spectral array (DSA) spectrograms displayed by Masimo SedLine – to guide sevoflurane titration, the amount needed to induce and maintain appropriate sedation could be reduced, thereby reducing the incidence of PAED.
In the study, children aged 1 to 6 years scheduled for elective surgery involving at least 30 minutes of general anesthesia at the largest children’s hospital in Japan were randomly assigned to an experimental group (n=91), whose anesthesia was guided by Masimo SedLine, or a control group (n=86), whose anesthesia was guided using standard practice, i.e. a fixed sevoflurane dosage of 1.0 MAC. The researchers recorded the amount of sevoflurane exposure for each patient and the time elapsed between important events including intubation, extubation, arrival and discharge from the PACU, and emergence from anesthesia. All patients were assessed for PAED by clinicians blinded to the treatment assignment, using a standard scale.
The researchers found that the incidence of PAED was significantly lower among patients in the SedLine group, affecting 21% of patients vs. 35% of patients in the control group (p=0.04). SedLine group patients were exposed to significantly less sevoflurane, by an average 1.4 MAC hours. They were extubated sooner, by an average of 3.3 minutes, regained consciousness sooner, by an average of 21.4 minutes, and were discharged from the PACU sooner, by an average of 16.5 minutes. The researchers estimated that the reduction in time spent in the OR and PACU, of approximately 20 minutes, may represent a cost savings of $240 to $920 (USD) per case.
The authors concluded, “EEG-guided management of general anesthesia reduced sevoflurane exposure and pediatric anesthesia emergence delirium in children, with faster emergence and shorter post-anesthesia care unit stays. The findings suggest that high concentrations of sevoflurane for induction followed by routine use of 1.0-MAC sevoflurane for maintenance may be excessive.”
They also noted that “with EEG monitoring, parents and guardians may be reassured that health care professionals can make an active effort to reduce and minimize a child’s exposure to anesthetic drugs.”
A similar study in 2022 by Long et al. – one of the first to investigate the impact of EEG-guided anesthesia on children undergoing surgery – found that pediatric patients monitored with SedLine experienced significantly fewer EEG patterns of profound brain inactivity, known as burst suppression. Burst Suppression has also been associated with adverse outcomes, including postoperative delirium.6
Yasuko Nagasaka, Professor of Anesthesia at Tokyo Women’s Medical University and the new study’s senior author, commented, “While general anesthesia is necessary for pediatric patients undergoing surgery, parents and guardians may express concerns about their child’s exposure to anesthetic drugs. We can now offer reassurance by explaining that modern medical technologies, such as EEG-guided anesthesia care, help minimize anesthetic exposure, potentially reducing the incidence of pediatric emergence delirium (PAED) and supporting earlier awakening and recovery from unconsciousness.”
“It is important to recognize,” continued Dr. Nagasaka, “that increasing the depth of anesthesia is relatively easy to learn. On the other hand, developing the confidence and skill to safely reduce anesthesia requires deeper understanding. Our results may offer a great step forward towards the improvement of our common practice, which may lead to the development of guidelines for EEG monitoring in pediatric areas to eventually mandate EEG monitoring during general anesthesia in the future. But comprehensive training in EEG-guided anesthesia should be considered a critical next step for the entire anesthesia community.”
Dean Kurth, MD, attending anesthesiologist at Children’s Hospital of Philadelphia and Professor of Anesthesiology and Critical Care Medicine at the University of Pennsylvania Perelman School of Medicine, added, “This study by Miyasaka et al. confirms the growing realization by pediatric anesthesiologists worldwide that more anesthesia drug is being administered to children than necessary, which has negative consequences. As the authors have shown, SedLine EEG data can help clinicians provide more precise anesthetic drug dosing for each child and improves outcomes.”
@Masimo | #Masimo
References
- Miyasaka K, Suzuki Y, Brown E, Nagasaka Y. EEG-Guided Titration of Sevoflurane and Pediatric Anesthesia Emergence Delirium. JAMA Pediatrics. April 21, 2025. DOI: 10.1001/jamapediatrics.2025.0517.
- Cole JW, Murray DJ, McAllister JD, Hirshberg GE. Emergence behavior in children: defining the incidence of excitement and agitation following anaesthesia. Paediatr Anaesth. 2002;12(5):442-447. DOI:10.1046/j.1460-9592.2002.00868.x.
- Davis L, Qi TS, and Ng A. Emergence delirium: an overview with an emphasis on the use of electroencephalography in its management. Anest Pain Med. 2024;19(Suppl 1):S87-95. DOI: 10.17085/apm.24013.
- Mason KP. Paediatric emergence delirium: a comprehensive review and interpretation of the literature. Br J Anaesth. 2017;118(3):335-343. DOI: 10.1093/bja/aew477.
- Goddard N, Smith D. Unintended awareness and monitoring of depth of anaesthesia. Contin Educ Anaesth Crit Care Pain. 2013;13(6):213-217.
- Long MHY, Lim EHL, Balanza GA, Allen JC, Purdon PL, and Bong CL. Sevoflurane requirements during electroencephalogram (EEG)-guided vs. standard anesthesia care in children: A randomized controlled trial. J Clin Anesth. 27 Jun 2022. DOI: https://doi.org/10.1016/j.jclinane.2022.110913.
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. Our mission is to improve life, improve patient outcomes, reduce the cost of care, and take noninvasive monitoring to new sites and applications. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown to outperform other pulse oximetry technologies in over 100 independent and objective studies, which can be found at www.masimo.com/evidence/featured-studies/feature. Masimo SET® is estimated to be used on more than 200 million patients around the world each year and is the primary pulse oximetry at all 10 top U.S. hospitals as ranked in the 2025 Newsweek World’s Best Hospitals listing. Additional information about Masimo and its products may be found at www.masimo.com.
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. Forward-looking statements are statements other than statements of historical facts that address activities, events or developments that we expect, believe or anticipate will or may occur in the future. These forward-looking statements include, among others, statements regarding the performance of SedLine to achieve certain results including the incidence of pediatric emergence delirium, earlier awakening, recovery from unconsciousness, or cost savings; the rate of adoption of SedLine technology by pediatric anesthesiologists for use during anesthesia administration in children undergoing surgery; and other matters that do not relate strictly to historical facts or statements of assumptions underlying any of the foregoing. These statements are often identified by the use of words such as “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “on-going,” “opportunity,” “plan,” “potential,” “predicts,” “forecast,” “project,” “seek,” “should,” “will,” or “would,” the negative versions of these terms and similar expressions or variations, but the absence of such words does not mean that a statement is not forward-looking. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: the highly competitive nature of the markets in which we sell our products and technologies; the ability to obtain regulatory approvals; the lack of acceptance of any of our current or future products and technologies; and other factors discussed in the “Risk Factors” section of our most recent periodic reports filed with the Securities and Exchange Commission (“SEC”), including our most recent Form 10-K and Form 10-Q, all of which you may obtain for free on the SEC’s website at www.sec.gov. Forward-looking statements are not guarantees of future performance. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today’s date. We do not undertake any obligation to update, amend or clarify these statements or the “Risk Factors” contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250610391732/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
Phase 3 Data for Incyte’s Retifanlimab (Zynyz®) in Patients with Squamous Cell Carcinoma of the Anal Canal (SCAC) Published in The Lancet13.6.2025 00:45:00 CEST | Press release
POD1UM-303/InterAACT 2 is the first and largest global Phase 3 trial evaluating a PD-1 inhibitor in combination with chemotherapy for the treatment of patients with advanced SCAC not previously treated with systemic chemotherapy The trial met its primary endpoint; treatment with retifanlimab in combination with platinum-based chemotherapy (carboplatin-paclitaxel) resulted in clinically meaningful improvements in progression-free survival and overall survival In May 2025, the U.S. Food and Drug Administration (FDA) approved Zynyz® (retifanlimab-dlwr) in combination with carboplatin and paclitaxel and as a single agent for the treatment of advanced SCAC; submissions to other global regulatory agencies are also under review Incyte (Nasdaq:INCY) today announced that primary results from the Phase 3 POD1UM-303/InterAACT 2 trial of retifanlimab (Zynyz®), a humanized monoclonal antibody targeting programmed death receptor-1 (PD-1), in combination with carboplatin and paclitaxel (platinum-base
Güntner Drives Group-Wide Innovation in 2024 Sustainability Report12.6.2025 21:30:00 CEST | Press release
Progress in natural refrigerants, water efficiency and circular economy underscores Güntner’s global impact Güntner, a global leader in refrigeration and heat exchange technology, today announced its featured role in the newly released 2024 A-HEAT Sustainability Report. The report outlines significant progress toward ambitious environmental goals—fueled by Güntner’s commitment to measurable, innovation-led sustainability. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250612897493/en/ Güntner advances global sustainability goals with progress in natural refrigerants, water savings, and circular economy efforts. Working toward its 2030 sustainability targets, Güntner delivered significant achievements across key areas including natural refrigeration integration, water and energy efficiency and the development of circular economy solutions. These efforts reflect Güntner’s ongoing mission to make sustainability not just a goal,
Windsurf Launches First GPU Cluster in Germany, Expanding Secure Enterprise AI in Europe12.6.2025 20:45:00 CEST | Press release
Windsurf launches a GPU cluster in Germany, delivering 50% faster performance and secure access to European enterprises. Data residency ensures enterprise repositories stay within the EU, meeting stringent security requirements. Windsurf already supports 100+ European enterprise customers, highlighting strong demand for AI-powered development tools across EMEA. Windsurf, a leader in AI-powered software development, today announced its first GPU cluster in Germany, as product demand surges and the company expands performance capacity across the EU. With over 100 local enterprise customers already using Windsurf products, the new cluster marks the company’s first major international step in building scalable infrastructure for the European market. Located in Frankfurt, home to some of the world’s strictest data security laws, the cluster was a natural choice for Windsurf’s first deployment. This cluster was specifically designed to meet the unique security and performance requirements of
Xsolla Partners With Defold to Enable Seamless In-game Purchases for Web Developers12.6.2025 19:00:00 CEST | Press release
New SDK Integration Simplifies Monetization for Defold Developers Publishing on CrazyGames Ahead of Global Game Jam Xsolla, a leading global video game commerce company, announces a new strategic partnership with Defold, the cross-platform game engine trusted by professionals and indie developers worldwide. This collaboration introduces a seamless integration between Xsolla and Defold-built web games on CrazyGames, enabling developers to easily monetize their titles just in time for the CrazyGames x Defold Game Jam running June 13–15. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250612569206/en/ (Graphic: Xsolla) To address the complexity of in-game monetization, Xsolla and Defold have developed a new SDK that allows Defold developers to implement in-game purchasing functionality without the complex manual API setup required. With Xsolla’s sponsorship and support, this integration removes friction from the monetization jou
Pioneering Cancer Plasticity Atlas will Help Predict Response to Cancer Therapies12.6.2025 17:00:00 CEST | Press release
The Wellcome Sanger Institute, Parse Biosciences, and the Computational Health Center at Helmholtz Munich today announced a collaboration to build the foundation of a single cell atlas, focused on understanding and elucidating cancer plasticity in response to therapies. The collaboration will catalyze an ambitious future phase to develop a cancer plasticity atlas encompassing hundreds of millions of cells. Utilizing novel organoid perturbation and Artificial Intelligence (AI) platforms, the aim is to create a comprehensive dataset to fuel foundational drug discovery models and cancer research. Dr. Mathew Garnett, Group Leader at the Sanger Institute, and Prof. Fabian Theis, Director of the Computational Health Center at Helmholtz Munich and Associate Faculty at the Sanger Institute, will be the principal investigators in the collaboration. Garnett’s research team has generated novel 3D organoid cultures that serve as highly scalable and functional cancer models with the ability to capt
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom