AIT Worldwide Logistics acquires Miami-based forwarder, GSDMIA, Inc.
22.4.2025 17:04:00 CEST | Business Wire | Press release
Strategic deal enhances trade lane support between Asia, Europe, Latin America, United States
Global supply chain solutions leader AIT Worldwide Logistics has finalized an agreement to purchase the assets of GSDMIA, Inc. (“GSDMIA”), an international freight forwarder based in Miami.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250422511696/en/
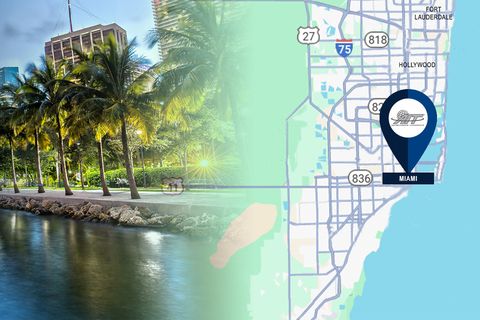
The agreement to purchase GSDMIA's assets boosts AIT's specialized service expertise for the cosmetics, industrial and technology sectors.
According to AIT’s Chief Development Officer, Ray Fennelly, who guided the acquisition process, GSDMIA provides deep international experience with air and ocean transportation. The team ships a diverse range of commodities between the United States, Asia, Europe and Latin America, including packaging and finished goods for the cosmetics industry, commercial HVAC equipment, and cryptocurrency data servers.
“GSDMIA is an extremely dynamic group that shares AIT’s focus on delivering a world-class customer experience,” Fennelly added. “I'm delighted to welcome the team to our network and look forward to working together to grow AIT’s reach and service reputation across the globe.”
After previously operating as an independent contractor with another large forwarder, GSDMIA’s co-founders, Wenzel Lewinsky and Joao Rios, arrive at AIT with nearly 60 years of combined global supply chain experience. Lewinsky and Rios have merged their business with the existing AIT office in Miami, assuming new roles as sales manager and station manager, respectively.
“AIT is a great fit, not only for our operation but, more importantly, for our customers,” Lewinsky said. “We’re looking forward to leveraging AIT's expansive global network and delivering even more efficient, high-quality solutions.”
“AIT’s scale of purchasing power, carrier relationships, and advanced shipping technology will allow us to take the tailored, value-added services we’re known for to an even higher level,” Rios added.
Terms of the acquisition have not been disclosed.
About AIT Worldwide Logistics
AIT Worldwide Logistics is a global freight forwarder that helps companies grow by expanding access to markets all over the world where they can sell and/or procure their raw materials, components and finished goods. For more than 45 years, the Chicago-based supply chain solutions leader has relied on a consultative approach to build a global network and trusted partnerships in nearly every industry, including aerospace, energy, high-tech, home delivery, life sciences, marine and more. Backed by scalable, user-friendly technology, AIT’s flexible business model customizes end-to-end deliveries via sea, air, road and rail — on time and on budget. With expert teammates staffing more than 150 worldwide locations in Asia, Europe, the Middle East and North America, AIT’s full-service options also include customs clearance, warehouse management and white glove services. Learn more at www.aitworldwide.com.
Our Mission
At AIT, we vigorously seek opportunities to earn our customers’ trust by delivering exceptional worldwide logistics solutions while passionately valuing our co-workers, partners and communities.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250422511696/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
Cosylab and Heron Neutron Medical Corp. Sign Letter of Intent to Advance Global Deployment of Accelerator-Based BNCT Systems10.6.2026 11:30:00 CEST | Press release
Cosylab and Heron Neutron Medical Corp. announced today, 10 June 2026, the signing of a Letter of Intent to establish a strategic framework for joint market development to support the global deployment of Accelerator-Based Boron Neutron Capture Therapy (AB-BNCT) systems. The collaboration aims to accelerate the development and integration of sophisticated software solutions for use with AB-BNCT while strengthening AB-BNCT's international market access and business development. By combining Cosylab's proven expertise in mission-critical software and complex system integration with Heron's end-to-end BNCT platforms, the partnership aims to drive broader global clinical adoption of this highly targeted cancer treatment modality. "AB-BNCT is emerging as one of the most promising approaches for treating difficult-to-treat and recurrent cancers," said Mark Pleško, Chief Executive Officer of Cosylab. "This collaboration reinforces our strategic focus on advancing radiation therapy technologie
GlobalFoundries and Qualinx Demonstrate First European Sovereign Manufacturing Flow for Security‑Critical Semiconductors10.6.2026 11:25:00 CEST | Press release
First end-to-end European chip manufacturing flow proven for aerospace, defence, IoT, consumer electronics and critical infrastructure GlobalFoundries (GF) and Qualinx today announced the successful completion of the first fully European-based, end-to-end semiconductor manufacturing flow at GlobalFoundries’ Dresden fab on its FDX technology. The milestone demonstrates that security-critical chips for aerospace, defence and critical infrastructure can be designed, manufactured and delivered entirely within Europe. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260610196220/en/ In this partnership, Qualinx served as the launch customer with a sophisticated GNSS SoC design for secure Positioning, Navigation and Timing (PNT) applications. The QLX3xx design targets sovereign GNSS-based PNT solutions for aerospace, defence and critical infrastructures—such as resilient timing and synchronization networks and highly integrated, ult
LTM Introduces BlueVerse™ Currency to Enable Outcome-Based Pricing in the Agentic AI Era10.6.2026 10:30:00 CEST | Press release
LTM, the Business Creativity partner to the world’s largest enterprises, today introduced BlueVerse™ Currency, an AI-linked commercial model that prices enterprise work by outcomes rather than effort consumed as enterprises scale agentic AI across core processes. BlueVerse Currency brings together the full BlueVerse stack—People (expertise on demand), Accelerators (reusable assets that speed delivery), Agents (autonomous AI workforce), Platforms (orchestration layer to scale delivery), and Tokens (compute and model usage)—into a single commercial construct. It introduces outcome-linked pricing tied to measurable business results, enabling shared productivity gains so clients can capture AI-driven efficiency value. The model also supports fungible resourcing, enabling reallocation mid-contract to evolving priorities, and faster reinvestment, with savings redirected into innovation. It is underpinned by a hybrid structure with fixed and variable pricing components, supported by use-case–
Smead Capital Management Extends International Value Strategy to Global Investors Through New Fund10.6.2026 09:00:00 CEST | Press release
Firm expands access to long-standing U.S.-based international strategy, bringing disciplined, value-driven investing to a broader global audience through the Smead Global ex-US Value UCITS Fund Smead Capital Management, a Phoenix-based investment management firm and a leader in value investing, today announced the launch of the Smead Global ex-US Value UCITS Fund designed to provide global investors with access to a product modeled after the firm’s long-standing Smead International Value Strategy. The Smead Global ex-US Value UCITS Fund is domiciled in Luxembourg. Investors can access the fund through Smead Capital Management’s website and it is available to professional and qualified investors through the fund’s transfer agent. The strategy that the Smead Global ex-US Value UCITS Fund is modeled after, the Smead International Value Strategy, has been a core contributor to Smead Capital Management’s outstanding performance for over a decade. Like all Smead products, the Global ex-US Va
Dodge Opens Orders for the Next Generation Charger in Europe10.6.2026 09:00:00 CEST | Press release
The iconic American muscle car returns with an all-electric and gasoline-powered lineup engineered without compromise Orders are now open in Europe for the next-generation Dodge Charger lineup The range includes the all-electric Dodge Charger Daytona and the gas-engine, SIXPACK-powered Dodge Charger Built on the STLA Large multi-energy platform, the new Charger delivers two distinct performance experiences rooted in the same Dodge DNA Available in both two-door and four-door body styles, with R/T and Scat Pack trims across both gasoline and all-electric versions European customers can order through KWA, Dodge’s official importer, using this link An Icon Enters Its Next Generation This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609386800/en/ Celebrating its 60th anniversary this year, Dodge opens a bold new chapter in Europe with the launch of the next-generation Dodge Charger, bringing one of America’s most iconic muscle c
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom