RevBio Initiates its Pivotal Clinical Trial in Europe for its Dental Implant Stabilization Product
17.3.2025 20:28:00 CET | Business Wire | Press release
The Company Received Approval in Multiple European Countries to Initiate this Key Study Necessary for Commercial Product Approval
RevBio, Inc., announced that it has received regulatory and ethics committee approvals in multiple European countries to conduct its pivotal clinical trial for its dental implant stabilization product. The successful completion of this pivotal clinical trial will result in the CE marking approval for the product, which will allow the company to begin commercial sales in Europe. As of the date of this press release, the company has already enrolled 30 of an expected 75 patients in this clinical trial.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250306978603/en/

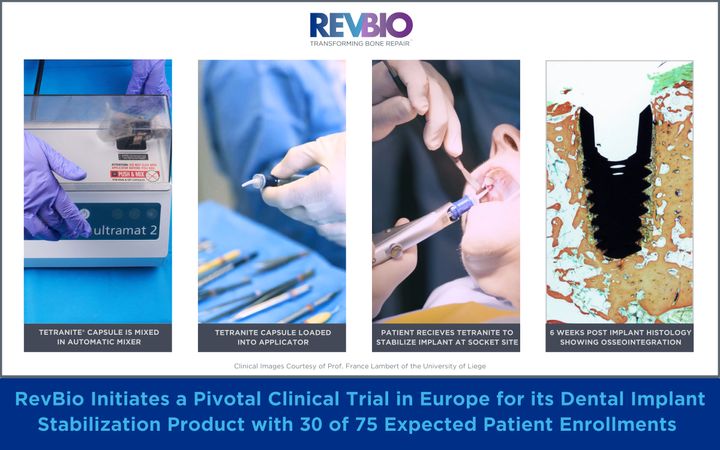
RevBio initiates a pivotal clinical trial in Europe for its Dental Implant Stabilization product with 30 of 75 expected patient enrollments. Clinical Images courtesy of Prof. France Lambert of the University of Liege.
“The results from this ongoing international multicenter study are exceeding expectations, showcasing remarkable outcomes. We are profoundly optimistic and incredibly enthusiastic about what the future holds,” said Prof. Dr. med. Dent., Patrick Schmidlin, head of the Division of Periodontology at the University of Zurich, who serves as one of the investigators in RevBio’s pivotal clinical trial. “This breakthrough material promises to open a new chapter in dental care, redefining what is possible in regenerative dentistry.”
RevBio received regulatory and ethics committee approvals for five clinical sites consisting of one in Switzerland, two in Belgium, one in Spain, and one in the United Kingdom. Each site is expected to enroll between 12-25 patients for a total of approximately 75 patients. The investigators who are approved to enroll patients in this clinical trial are Prof. Dr. med. Dent., PhD, Patrick Schmidlin, head of the Department of Periodontology at the University of Zurich, Ana Castro, BDS MSc PhD, Clinical Head Department Periodontology at the Catholic University of Leuven, France Lambert, DDS, PhD, Professor and Head of Periodontology, Oral Surgery, and Implant Surgery at the University of Liege, Arturo Llobell, DDS, MS, a periodontist in private practice in Valencia, Spain, and Azim Malik, BDS, MFDS RCSEd, DipPCD RCSI, DClinDent, MPerio RCSEd, a periodontist and implant specialist in private practice in London, United Kingdom.
When teeth are extracted due to damage from traumatic injuries, tooth decay, or gum disease, the current standard of care consists of multiple staged surgical procedures to restore a patient’s dentition with prosthetic crowns supported by dental implants. Frequently, extraction sites are too large for dental implants to achieve primary stability through conventional mechanical engagement. Instead, patients must undergo a costly, complex, and lengthy process including a preliminary bone grafting surgery before receiving a dental implant. The use of TETRANITE to stabilize an unstable implant will allow for the immediate placement of dental implants which otherwise could not be placed until the initial bone graft has healed to form new bone. As a result, the TETRANITE® biomaterial will help reduce the duration and complexity of these dental implant procedures, lessen patient pain and recovery time, and reduce the overall cost of care thereby providing greater patient access for the treatment of tooth loss.
"The approval to conduct this pivotal clinical trial and the successful enrollment of the first 30 patients is a watershed moment for RevBio," said Alan Pollack, DDS, RevBio’s Senior Director of Dental Clinical Operations. “This clinical trial has the potential to significantly accelerate the timeframe when this promising bone adhesive technology can be used in the field of dentistry.”
About RevBio, Inc.
RevBio, Inc., is a clinical stage medical device company engaged in the development and commercialization of TETRANITE®, a patented, synthetic, injectable, self-setting, and osteoconductive bone adhesive biomaterial. The company is initially developing this technology for use in the dental, cranial, and broader orthopaedic markets as well as applications in the animal health market. RevBio's TETRANITE technology is not yet approved for commercial use.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250306978603/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
Blue Planet Prize 2026: Announcement of Prize Laureates10.6.2026 05:00:00 CEST | Press release
This year marks the 35th awarding of the Blue Planet Prize, the international environmental award sponsored by the Asahi Glass Foundation, chaired by Takuya Shimamura. Every year, the Foundation selects two laureates, individuals, or organizations who have made significant contributions towards the resolution of global environmental problems. The Board of Directors has selected the following 2026 Blue Planet Prize laureates. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260603774361/en/ Dr. Linda S. Birnbaum (USA), Professor Edward Barbier (USA) 1. Dr. Linda S. Birnbaum (USA) born December 21, 1946 Former Director of the National Institute of Environmental Health Sciences (NIEHS) Former Director of the National Toxicology Program (NTP) Dr. Linda S. Birnbaum led pioneering research on the toxicity of persistent organic pollutants (POPs), significantly advancing international risk assessment. By demonstrating how exposure to
NIQ (NYSE: NIQ) Unveils Six New AI-Powered Capabilities at C360, Executing on Its Vision for Agentic Commerce9.6.2026 21:00:00 CEST | Press release
New capabilities help organizations operationalize trusted market intelligence across enterprise AI environments and workflows, while preparing for the future of agentic commerce NielsenIQ (NYSE: NIQ) today unveiled six new AI-powered capabilities at C360, the company's flagship client and industry event, marking a major milestone in the execution of NIQ’s AI strategy. Thenew capabilities help brands, retailers, and other organizations operationalize trusted market intelligence across enterprise workflows, AI environments, and the emerging world of agentic commerce. NIQ’s AI value creation strategy is built on two foundational principles: Delivering The Full View™: Combining the industry’s broadest and deepest datasets, advanced AI models, and rich content to help organizations unlock actionable insights and drive AI innovation at scale. Enabling AI-Powered Capabilities: Delivering trusted intelligence and proprietary IP through NIQ’s advanced AI applications, assistants, and agents em
H55 Delivers Certification-Grade Propulsion Battery Modules to Pratt & Whitney Canada, Supporting Demonstration of Hybrid-Electric Aircraft Technology9.6.2026 17:31:00 CEST | Press release
Production-conforming systems manufactured in a regulator-approved facility mark H55’s transition from technology development to commercialization of certification-grade energy storage solutions for hybrid-electric aviation. H55 today announced the delivery of certification-grade propulsion battery modules to Pratt & Whitney Canada in support of the RTX Hybrid-Electric Flight Demonstrator — a milestone that further validates H55’s transition from technology development to industrial-scale execution and represents an important step in the commercialization of the company’s certification-grade energy storage technologies for hybrid-electric aerospace applications. Pratt & Whitney is an RTX business. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609825006/en/ H55 Adagio Battery Modules in Pratt & Whitney Canada Hangar ©RTX The delivery represents more than hardware integration. It demonstrates H55’s ability to manufacture p
New Polymorph of Indomethacin Discovered9.6.2026 17:00:00 CEST | Press release
- Joint Research Involving Rigaku Published in Crystal Growth & Design - Rigaku Corporation, a global solution partner in X-ray analytical systems and a group company of Rigaku Holdings Corporation (headquarters: Akishima, Tokyo; CEO: Jun Kawakami; “Rigaku”), announced that the results of a joint research project conducted with Shionogi & Co., Ltd., JEOL Ltd., and Meiji Pharmaceutical University have been published in Crystal Growth & Design, a world-renowned international journal in the field of crystallography. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609347405/en/ Cover of the June 2026 issue of Crystal Growth & Design, featuring this research. This research uncovered a previously unknown polymorph (κ-form) of indomethacin, a widely used pain relief and anti-inflammatory drug. The research team also conducted structural analysis and characterization of the newly identified crystal form. Although indomethacin has
SLB Collaborates with Qualcomm on Edge AI Solutions for Energy Operations9.6.2026 16:28:00 CEST | Press release
Collaboration combines edge computing and energy workflows to support real-time operational decision-making Global energy technology company SLB (NYSE: SLB) today announced a memorandum of understanding with Qualcomm Technologies, Inc. to enable edge AI solutions for the energy industry, supporting real-time operational decision-making across wells, facilities and production systems. The collaboration combines Qualcomm Technologies’ low-power edge computing and AI processing capabilities, with SLB’s Agora™ edge AI and IoT solutions developed for remote and operationally complex environments. “Together, SLB and Qualcomm Technologies aim to help operators apply AI more effectively across energy infrastructure,” said Rakesh Jaggi, president, Digital, SLB. “Many energy operations rely on real-time decision-making in remote environments where connectivity and responsiveness directly affect performance. AI systems designed around the realities of energy operations can help support more consi
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom