Net Profit of Estithmar Holding Increases by 20% to QAR 422 Million, Revenues Rise by 44% to Reach QAR 4.2 Billion
13.2.2025 18:13:00 CET | Business Wire | Press release
Financial Results Announcement for the FY 2024
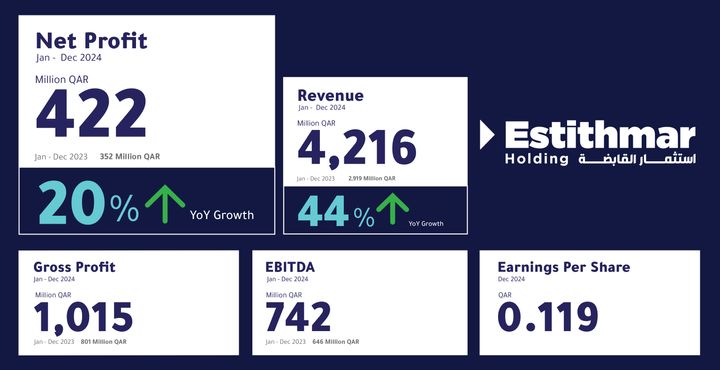
Estithmar Holding Q.P.S.C. announced its financial results for the year ended December 31, 2024, reporting a 44% increase in revenues, reaching QAR 4.2 billion, compared to QAR 2.9 billion in 2023. The gross profits of the company reached QAR 1 billion, up from QAR 801 million in 2023, reflecting a 27% increase. The company also achieved earnings before interest, tax, depreciation, and amortization (EBITDA) of QAR 742 million. The results showed a 20% rise in the company’s net profit compared to 2023, reaching QAR 422 million. Earnings per share also increased by 17% from 2023, reaching QAR 0.119 per share.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250213715874/en/
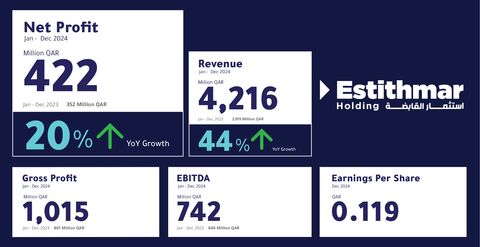
Net Profit of Estithmar Holding Increases by 20% to QAR 422 Million, Revenues Rise by 44% to Reach QAR 4.2 Billion (Graphic: AETOSWire)
The 20% growth in net profit is attributed to the increase in the company’s revenues, which were primarily driven by the contracting and healthcare clusters. The contracting cluster expanded in executing and delivering projects efficiently in the Kingdom of Saudi Arabia. The healthcare cluster contributed to the rise in 2024 revenues, driven by the sustainable performance growth of The View Hospital in Qatar, and the expansion of the cluster through the subsidiary Apex Health, which signed management and operation agreements for several hospitals. A set of measures taken in the ventures cluster has also contributed to restructuring the business model and increasing revenues.
The financial results for 2024 reported a 25% increase in assets, resulting from the new projects added to Estithmar’s portfolio. The results also revealed a current liquidity ratio of 1.22.
The Board of Directors of Estithmar Holding recommended a dividend distribution equivalent to 10% of the capital, with 1 free share for every 10 shares.
For more information: Estithmar Holding - Legacy of Excellence.
*Source: AETOSWire
View source version on businesswire.com: https://www.businesswire.com/news/home/20250213715874/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
H55 Delivers Certification-Grade Propulsion Battery Modules to Pratt & Whitney Canada, Supporting Demonstration of Hybrid-Electric Aircraft Technology9.6.2026 17:31:00 CEST | Press release
Production-conforming systems manufactured in a regulator-approved facility mark H55’s transition from technology development to commercialization of certification-grade energy storage solutions for hybrid-electric aviation. H55 today announced the delivery of certification-grade propulsion battery modules to Pratt & Whitney Canada in support of the RTX Hybrid-Electric Flight Demonstrator — a milestone that further validates H55’s transition from technology development to industrial-scale execution and represents an important step in the commercialization of the company’s certification-grade energy storage technologies for hybrid-electric aerospace applications. Pratt & Whitney is an RTX business. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609825006/en/ H55 Adagio Battery Modules in Pratt & Whitney Canada Hangar ©RTX The delivery represents more than hardware integration. It demonstrates H55’s ability to manufacture p
New Polymorph of Indomethacin Discovered9.6.2026 17:00:00 CEST | Press release
- Joint Research Involving Rigaku Published in Crystal Growth & Design - Rigaku Corporation, a global solution partner in X-ray analytical systems and a group company of Rigaku Holdings Corporation (headquarters: Akishima, Tokyo; CEO: Jun Kawakami; “Rigaku”), announced that the results of a joint research project conducted with Shionogi & Co., Ltd., JEOL Ltd., and Meiji Pharmaceutical University have been published in Crystal Growth & Design, a world-renowned international journal in the field of crystallography. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609347405/en/ Cover of the June 2026 issue of Crystal Growth & Design, featuring this research. This research uncovered a previously unknown polymorph (κ-form) of indomethacin, a widely used pain relief and anti-inflammatory drug. The research team also conducted structural analysis and characterization of the newly identified crystal form. Although indomethacin has
SLB Collaborates with Qualcomm on Edge AI Solutions for Energy Operations9.6.2026 16:28:00 CEST | Press release
Collaboration combines edge computing and energy workflows to support real-time operational decision-making Global energy technology company SLB (NYSE: SLB) today announced a memorandum of understanding with Qualcomm Technologies, Inc. to enable edge AI solutions for the energy industry, supporting real-time operational decision-making across wells, facilities and production systems. The collaboration combines Qualcomm Technologies’ low-power edge computing and AI processing capabilities, with SLB’s Agora™ edge AI and IoT solutions developed for remote and operationally complex environments. “Together, SLB and Qualcomm Technologies aim to help operators apply AI more effectively across energy infrastructure,” said Rakesh Jaggi, president, Digital, SLB. “Many energy operations rely on real-time decision-making in remote environments where connectivity and responsiveness directly affect performance. AI systems designed around the realities of energy operations can help support more consi
RevBits and Stony Brook University’s Ethos Lab Establish a Collaborative Partnership to Further the Field of Cyber Security Education and Application9.6.2026 16:15:00 CEST | Press release
Through the partnership, RevBits will provide its full suite of cybersecurity solutions to Stony Brook University’s Ethos Lab, to deliver the capability to advance student education and equip them with the knowledge needed to face modern cyber threats in a realistic, contained environment. Stony Brook University’s (SBU) Ethos Lab, in the College of Engineering and Applied Sciences Department of Computer Science, recently announced a collaborative partnership with RevBits, LLC. Through the partnership, SBU’s Ethos Lab will utilize the RevBits solutions suite to build computer science labs and a cybersecurity-focused curriculum, reinforcing lessons from the classroom. The platform simulates environments containing five major threat landscapes, including Endpoint Security, Privileged Access Management, Email Security, Zero Trust Network and Deception Technology. SBU’s Department of Computer Science was recently designated as a National Center of Academic Excellence in Cybersecurity Resear
Marie® by Leo Cancer Care Makes History at Stanford Medicine — World First in Compact Upright Proton Therapy9.6.2026 15:22:00 CEST | Press release
The first patient treated was a child. The milestone marks the moment upright proton therapy moves from innovation to adoption — and reflects exactly what Leo Cancer Care built Marie® to achieve Following the landmark first treatment at Stanford Medicine Cancer Center Care June 4, Leo Cancer Care today announces the role of its Marie® upright patient positioning and imaging platform in enabling the world's first compact upright proton therapy patient treatment. Delivered using the Mevion S250-FIT™ Proton Therapy System and powered by RayStation from RaySearch, the milestone is the culmination of a decade of development. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609870736/en/ The treatment room at Stanford Medicine Cancer Center, Palo Alto, California, housing the Marie® upright patient positioning platform by Leo Cancer Care (center foreground) integrated with the Mevion S250-FIT™ Proton Therapy System. The circular
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom