Regula Launches Full Support for Digital Travel Credentials in Latest Software Update
15.1.2025 08:00:00 CET | Business Wire | Press release
Regula, a global developer of forensic devices and identity verification solutions, has updated its Regula Document Reader SDK. Now, the software fully supports the new Digital Travel Credential (DTC) format, aligned with the International Civil Aviation Organization (ICAO) standards. This enhancement enables governments, airlines, and border control authorities worldwide to process travel documents with unmatched security, efficiency, and ease—whether on-site or remotely.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250115990355/en/
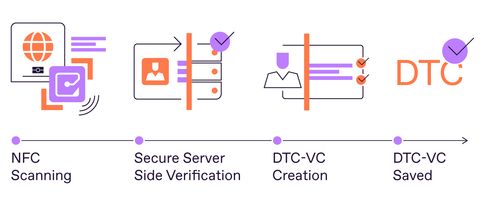
With Regula Document Reader SDK, travelers can create their own digital credentials by extracting a Virtual Component (DTC-VC) from a physical electronic identity document like ePassport. (Graphic: Regula)
The DTC is a secure digital solution that streamlines travel experiences. It consolidates key personal information into a single virtual document that travelers can store on their mobile devices or upload to their digital wallets and share whenever needed. The main goal of the DTC is to facilitate clearance procedures during travel and ensure that people are eligible to enter their destination before they board a flight.
The ICAO has defined three types of DTC, each offering varying levels of convenience and security for travelers.
- DTC Type 1 allows travelers to create their own digital credentials by extracting a Virtual Component (DTC-VC) from a physical electronic identity document, stored on their personal device. However, travelers must still carry the original document for identity verification.
- DTC Type 2, issued by authorities, combines a cryptographically linked Virtual Component (DTC-VC) and a Physical Component (DTC-PC). This format adds an extra layer of security while maintaining a connection to the physical document.
- DTC Type 3 represents the future of travel identification: a fully digital document issued directly by authorities. Unlike the other types, it eliminates the need for a physical ID, streamlining identity verification for a completely digital experience.
Now, with Regula Document Reader SDK, users can create and reprocess DTC-VC from ePassports, and verify it by passing DTC-VC data as input. Also, support for handling DTC-PC has been introduced. The updated Regula Document Reader SDK can:
- read the document’s RFID chip with a smartphone or passport reader and create DTC-VC;
- recognize, read, and verify DTC-VC with a smartphone, passport reader, or server;
- read DTC-PC with a smartphone or passport reader, parse its data, and verify it.
The updated Regula Document Reader SDK is equipped with advanced features that fully support DTC implementation.
- Trustworthy NFC verification. First and foremost, it provides trusted server-side NFC verification of the ID so it ensures accurate and trustworthy DTC-VC creation. Since all the data from the chip can be verified on a secure server, there is no need to question the reliability of the checks performed by a mobile device (which is prone to manipulation). Such an approach ensures that the virtual component of a traveler’s document is secure and taken from an authentic ID.
- Compliance with ICAO guidelines and technical reports. Regula Document Reader SDK not only verifies DTCs but also guarantees that each DTC fully complies with ICAO guidelines and technical reports. This makes Regula’s solution an indispensable tool for airlines and governments so they can be confident in the validity of travelers’ DTCs.
- Future-ready technology. In addition to supporting DTC-VC, Regula’s technology is fully compatible with handling DTC-PC. Looking further ahead, Regula is ready to process DTC Type 3, a digital passport that is expected in the next several years.
To facilitate the fast and smooth global application of DTCs, Regula Document Reader SDK relies on the most comprehensive identity document template database, which is owned and maintained by Regula. Currently, it contains more than 14,000 ID templates from 251 countries and territories, and it’s constantly growing.
Regula Document Reader SDK is designed for easy integration into third-party applications, allowing clients to incorporate this advanced DTC-ready technology seamlessly into their existing systems. With customizable options, businesses can adapt the solution to meet their unique operational requirements while providing end users with a secure, streamlined digital experience.
“We see that the world is rapidly moving to embrace digital IDs. According to the Forrester Consulting study commissioned by Regula earlier in 2024, nearly half of businesses around the world, 42%, are actively integrating digital IDs into their systems. For the Aviation sector, this rate is even higher: 50%. By ensuring full DTC support in the latest update of Regula Document Reader SDK, we are helping our clients to smoothly transition to the future of travel, where secure, digital-first solutions redefine the passenger experience,” says Ihar Kliashchou, Chief Technology Officer at Regula.
To learn more about the capabilities of the updated Regula Document Reader SDK, read the release documentation on Regula’s website.
About Regula
Regula is a global developer of forensic devices and identity verification solutions. With our 30+ years of experience in forensic research and the most comprehensive library of document templates in the world, we create breakthrough technologies for document and biometric verification. Our hardware and software solutions allow over 1,000 organizations and 80 border control authorities globally to provide top-notch client service without compromising safety, security, or speed. Regula has been repeatedly named a Representative Vendor in the Gartner® Market Guide for Identity Verification. Learn more at www.regulaforensics.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250115990355/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
NIQ (NYSE: NIQ) Unveils Six New AI-Powered Capabilities at C360, Executing on Its Vision for Agentic Commerce9.6.2026 21:00:00 CEST | Press release
New capabilities help organizations operationalize trusted market intelligence across enterprise AI environments and workflows, while preparing for the future of agentic commerce NielsenIQ (NYSE: NIQ) today unveiled six new AI-powered capabilities at C360, the company's flagship client and industry event, marking a major milestone in the execution of NIQ’s AI strategy. Thenew capabilities help brands, retailers, and other organizations operationalize trusted market intelligence across enterprise workflows, AI environments, and the emerging world of agentic commerce. NIQ’s AI value creation strategy is built on two foundational principles: Delivering The Full View™: Combining the industry’s broadest and deepest datasets, advanced AI models, and rich content to help organizations unlock actionable insights and drive AI innovation at scale. Enabling AI-Powered Capabilities: Delivering trusted intelligence and proprietary IP through NIQ’s advanced AI applications, assistants, and agents em
H55 Delivers Certification-Grade Propulsion Battery Modules to Pratt & Whitney Canada, Supporting Demonstration of Hybrid-Electric Aircraft Technology9.6.2026 17:31:00 CEST | Press release
Production-conforming systems manufactured in a regulator-approved facility mark H55’s transition from technology development to commercialization of certification-grade energy storage solutions for hybrid-electric aviation. H55 today announced the delivery of certification-grade propulsion battery modules to Pratt & Whitney Canada in support of the RTX Hybrid-Electric Flight Demonstrator — a milestone that further validates H55’s transition from technology development to industrial-scale execution and represents an important step in the commercialization of the company’s certification-grade energy storage technologies for hybrid-electric aerospace applications. Pratt & Whitney is an RTX business. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609825006/en/ H55 Adagio Battery Modules in Pratt & Whitney Canada Hangar ©RTX The delivery represents more than hardware integration. It demonstrates H55’s ability to manufacture p
New Polymorph of Indomethacin Discovered9.6.2026 17:00:00 CEST | Press release
- Joint Research Involving Rigaku Published in Crystal Growth & Design - Rigaku Corporation, a global solution partner in X-ray analytical systems and a group company of Rigaku Holdings Corporation (headquarters: Akishima, Tokyo; CEO: Jun Kawakami; “Rigaku”), announced that the results of a joint research project conducted with Shionogi & Co., Ltd., JEOL Ltd., and Meiji Pharmaceutical University have been published in Crystal Growth & Design, a world-renowned international journal in the field of crystallography. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609347405/en/ Cover of the June 2026 issue of Crystal Growth & Design, featuring this research. This research uncovered a previously unknown polymorph (κ-form) of indomethacin, a widely used pain relief and anti-inflammatory drug. The research team also conducted structural analysis and characterization of the newly identified crystal form. Although indomethacin has
SLB Collaborates with Qualcomm on Edge AI Solutions for Energy Operations9.6.2026 16:28:00 CEST | Press release
Collaboration combines edge computing and energy workflows to support real-time operational decision-making Global energy technology company SLB (NYSE: SLB) today announced a memorandum of understanding with Qualcomm Technologies, Inc. to enable edge AI solutions for the energy industry, supporting real-time operational decision-making across wells, facilities and production systems. The collaboration combines Qualcomm Technologies’ low-power edge computing and AI processing capabilities, with SLB’s Agora™ edge AI and IoT solutions developed for remote and operationally complex environments. “Together, SLB and Qualcomm Technologies aim to help operators apply AI more effectively across energy infrastructure,” said Rakesh Jaggi, president, Digital, SLB. “Many energy operations rely on real-time decision-making in remote environments where connectivity and responsiveness directly affect performance. AI systems designed around the realities of energy operations can help support more consi
RevBits and Stony Brook University’s Ethos Lab Establish a Collaborative Partnership to Further the Field of Cyber Security Education and Application9.6.2026 16:15:00 CEST | Press release
Through the partnership, RevBits will provide its full suite of cybersecurity solutions to Stony Brook University’s Ethos Lab, to deliver the capability to advance student education and equip them with the knowledge needed to face modern cyber threats in a realistic, contained environment. Stony Brook University’s (SBU) Ethos Lab, in the College of Engineering and Applied Sciences Department of Computer Science, recently announced a collaborative partnership with RevBits, LLC. Through the partnership, SBU’s Ethos Lab will utilize the RevBits solutions suite to build computer science labs and a cybersecurity-focused curriculum, reinforcing lessons from the classroom. The platform simulates environments containing five major threat landscapes, including Endpoint Security, Privileged Access Management, Email Security, Zero Trust Network and Deception Technology. SBU’s Department of Computer Science was recently designated as a National Center of Academic Excellence in Cybersecurity Resear
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom