SamanTree Medical Highlights Research Showing 67% Reduction in Reoperation Rates for Breast-Conserving Surgery at the 2024 SABCS
11.12.2024 15:00:00 CET | Business Wire | Press release
SHIELD study shows reoperation rates dropped from 30% to 10% with use of the Histolog® Scanner, highlighting its potential to improve oncologic surgery and patient outcomes
SamanTree Medical, a leader in oncologic surgical imaging innovation, today announced promising results from the SHIELD study, which will be presented by researchers on Dec. 12 at the 2024 San Antonio Breast Cancer Symposium (SABCS). The findings show that the Histolog® Scanner, a confocal laser microscope, reduced reoperation rates during breast-conserving surgery (BCS) from 30% to 10%, representing a 67% reduction. The innovative imaging device also demonstrated significant accuracy in detecting positive margins with high sensitivity and specificity.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20241211957581/en/
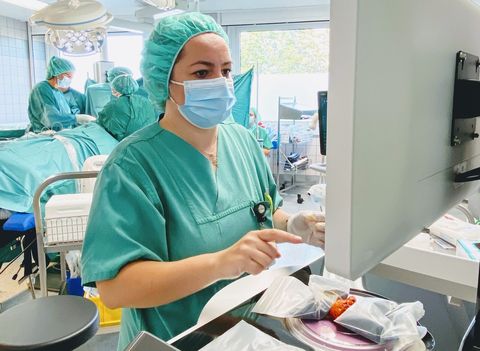
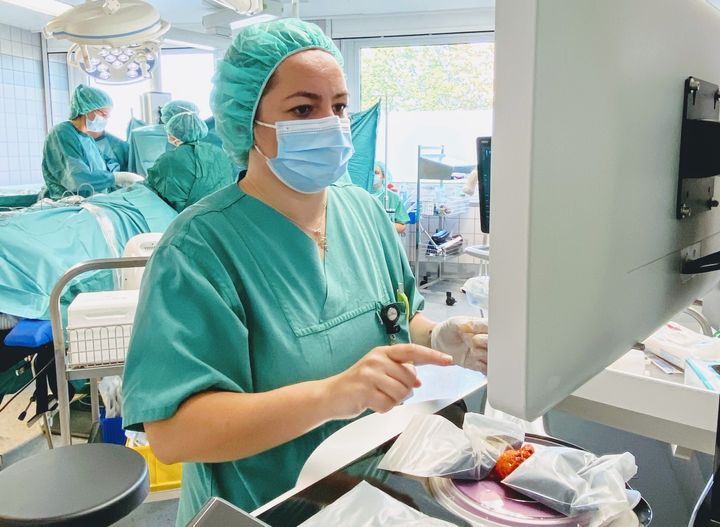
Dr. Mariana-Felicia Sandor, first author of the SHIELD study: Reoperation Rate in Breast-Conserving Surgery using Confocal Histolog® Scanner for Intraoperative Tissue Assessment, performs real-time, high-resolution tissue assessments during breast-conserving surgery (BCS) using the FDA-cleared Histolog Scanner. SHIELD study data indicates the innovative imaging device reduced reoperation rates during BCS from 30% to 10%, representing a 67% reduction. (Photo: Business Wire)
“The results of the SHIELD study impressively demonstrate a proof-of-concept of the Histolog Scanner in patients with breast cancer and also confirm the non-interventional POLARHIS study,” said Professor Michael Patrick Lux, M.D., principal investigator of the SHIELD study and Head of the Department of Gynecology and Obstetrics at St. Vincenz Hospital, Paderborn, Germany. “The data are absolutely relevant for clinical care, as the system can reduce the re-section rate in a clinically relevant and significant way, thus optimizing oncological safety, cosmetics, and the economic aspects of care.”
The prospective study, conducted at St. Vincenz Hospital, evaluated the impact of the Histolog Scanner on reducing reoperation rates during BCS. Fifty patients with invasive breast cancer and/or ductal carcinoma in situ (DCIS) were enrolled, and reoperation rates were compared to historical data from the POLARHIS study, a retrospective observational study performed at the same breast center by the same surgical team using standard intraoperative margin assessment techniques.
The results showed a dramatic reduction in reoperation rates from 30% in the POLARHIS study to 10% in the SHIELD study, representing a 67% improvement when the Histolog Scanner was used. Moreover, the Histolog Scanner outperformed standard techniques by achieving a sensitivity of 80.9% and a specificity of 99.5% for positive margin detection, compared to the combined sensitivity of 17.4% and specificity of 97.3% achieved with standard methods, as seen in the SHIELD study.
"These outstanding results underscore the potential of the Histolog Scanner to enhance oncologic surgical outcomes significantly," said Olivier Delporte, CEO of SamanTree Medical. "By enabling real-time, high-resolution tissue microstructure assessments during BCS, our technology addresses a critical unmet need, reducing reoperation rates and improving patient outcomes.
"With the recent FDA clearance, we are pleased to make this groundbreaking technology available to surgeons throughout the U.S. The Histolog Scanner sets a new standard for precision and care in oncologic surgery, marking a significant milestone in our mission to transform oncologic surgery and improve patient care globally," continued Delporte.
About SamanTree Medical
Headquartered in Liège, Belgium, with U.S. regional headquarters in Boston, SamanTree Medical SA is dedicated to improving oncologic surgery through innovative imaging solutions. Its flagship product, the Swiss-developed Histolog Scanner, features massive parallel confocal microscopy, a cutting-edge technology that expands the imaging field up to 40,000 times larger than standard confocal microscopy. The breakthrough device enables surgeons and pathologists to visualize large, fresh tissue samples in real-time with exceptional resolution and accuracy, making it uniquely suited for intraoperative environments in oncologic surgery. Founded in 2010 based on innovations from the École Polytechnique Fédérale de Lausanne (EPFL), a leading Swiss research institute renowned for engineering and technological advancements, SamanTree Medical is committed to improving surgical precision and outcomes by enabling real-time fresh tissue imaging. More information may be found here.
Click here for Important Safety Information.
View source version on businesswire.com: https://www.businesswire.com/news/home/20241211957581/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
Pure Lithium Corporation Named World Economic Forum Technology Pioneer10.6.2026 20:10:00 CEST | Press release
Lithium metal battery innovator joins a select global cohort of 100 pioneering companies shaping the future of energy and advanced materials Pure Lithium Corporation, a disruptive lithium metal battery technology company, is pleased to announce that the company has been selected as a 2026 Technology Pioneer by the World Economic Forum. Launched in 2000, the Technology Pioneers is a leading community for companies from around the world that are shaping the future through breakthrough technologies and innovations. These companies are selected for their potential to have a significant impact on business and society and are invited to engage with public and private sector leaders through the World Economic Forum’s global platform. “We are delighted to be selected as a World Economic Forum Technology Pioneer. I would like to express my gratitude to the World Economic Forum for this honor,” said Pure Lithium Founder, Chairman and CEO, Emilie Bodoin. “Pure Lithium’s selection is a testament t
Visa Partners with OpenAI to Power the Next Generation of AI Commerce10.6.2026 20:00:00 CEST | Press release
New collaboration brings Visa’s global payment networkto one of the largest AI platforms and aims to support seamless, secure transactions and broader AI-powered use cases across commerce environments supported by agents Visa (NYSE: V) today announced a strategic collaboration with OpenAI to enable secure Visa payments within agentic commerce, enabling seamless and trusted payments across OpenAI. The companies made the announcement at the Visa Payments Forum in San Francisco. Through the partnership, Visa will provide its global network, credentialing capabilities and security infrastructure to support agentic commerce experiences, helping consumers and businesses interact and transact with confidence. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260610422687/en/ The collaboration is part of the broader Visa Intelligent Commerce initiative, which is focused on extending secure payment capabilities into new digital environm
Visa Announces New AI, Stablecoin and Token Innovations to Power Intelligent, Programmable Commerce at Visa Payments Forum10.6.2026 20:00:00 CEST | Press release
New Agent Scoring, Agentic Registry and Large Transaction Model capabilities, stablecoin settlement and token enhancements support the next gen of digital commerce At Visa Payments Forum 2026, Visa (NYSE: V) today announced new AI, stablecoin and token capabilities designed to help clients unlock the next generation of commerce. These innovations reflect a simple objective: ensuring trust, security and control evolve alongside increasingly fast, automated and intelligent commerce experiences. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260610464331/en/ In a keynote presentation, Jack Forestell, Chief Product & Strategy Officer at Visa, outlined how two foundational shifts—artificial intelligence and stablecoins—are transforming both the front end and back end of money movement, and how Visa is enabling clients to participate. “AI is transforming the front end of commerce. Stablecoins are reshaping the back end,” said Fore
NEURA Robotics Announces Record Series C of up to $1.4 Billion to Accelerate the World’s Leading Physical AI Platform10.6.2026 17:26:00 CEST | Press release
Landmark investment drives NEURA's open ecosystem, where humanoid and cognitive robots share intelligence and capabilities at scaleBacking from Tether, Qualcomm Technologies, Amazon, NVIDIA, imec.xpand, Bosch, Schaeffler, European Investment Bank, Lingotto Horizon, InterAlpen Partners and others demonstrates global confidence in NEURA’s leadershipNew capital raise is the largest of a full-stack robotics company everCapital will drive serial production to multi-million robots by 2030 and support the global rollout of NEURA Gyms, the world’s first real-world training environments for cognitive robots and Physical AI NEURA Robotics (“NEURA”), the pioneer in cognitive robotics and creator of the Neuraverse, today announced a landmark Series C financing with a total round size of up to $1.4 billion to accelerate its mission of building the world’s leading Physical AI platform. The financing brings together global leaders across AI, robotics, compute, manufacturing and industrial infrastruct
ees Europe: Green Hydrogen: Opportunities and Production Ramp Up10.6.2026 16:13:00 CEST | Press release
For the major economies, the transformation towards a climate-neutral industry is one of the central tasks for the coming decades. While the European Union has pledged climate neutrality by 2050, China’s goal is to be climate-neutral by 2060. Hydrogen is a key to transforming their manufacturing and energy industry. However, the actual market development is lagging behind these ambitious objectives: According to the Global Energy Report 2025, only 11 percent of hydrogen projects announced since the start of the decade are now in operation or have been secured by investment decisions. This is why ees Europe, Europe’s largest and most international exhibition for batteries and energy storage systems, is putting a spotlight on hydrogen. The newly aligned exhibition segment Hydrogen Dialogue Forum & Expo in hall B2 will be giving in-depth information about the opportunities for hydrogen technology, technological innovation, savings potential and ground-breaking projects from June 23–25. Th
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom