Orthogon Therapeutics Closes Oversubscribed $5.2M Financing to Advance First Oral Therapy for BK Polyomavirus
10.12.2024 13:57:00 CET | Business Wire | Press release
Orthogon’s small molecule therapeutics redefine treatment possibilities for BK virus infections.
Orthogon Therapeutics, a developer of novel antiviral medicines, today announced the closing of an oversubscribed financing round, exceeding its $5M target and bringing total funding to over $25M. This funding will accelerate the development of its first-in-class treatments for managing BK virus reactivation in transplant patients.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20241210996847/en/
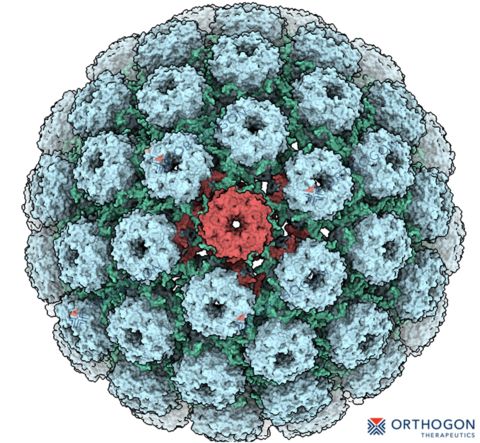
Ultrastructural representation of the BK polyomavirus capsid, featuring 72 pentamers that form its distinct icosahedral symmetry. (Graphic: Business Wire)
The company has successfully developed small molecule drugs that directly target viral proteins essential to the polyomavirus life cycle. Polyomaviruses lack conventional antiviral protein targets and have eluded drug development for decades. Backed by a diversified investor base and a pipeline of drugs with unique mechanisms of action, Orthogon is addressing this area of urgent unmet medical need.
“Our high-affinity small molecule antivirals rival the potency of biologics without their drawbacks,” said Dr. Stephen Weeks, VP of Structural Biology at Orthogon Therapeutics. “This achievement highlights our platform’s ability to target previously undruggable viral proteins.”
This breakthrough coincides with Orthogon’s creation of a pioneering animal model to study BK polyomavirus pathology. Historically, researchers have lacked in vivo insights into BK virus disease progression. This model deepens understanding of disease mechanisms and provides a robust platform for testing therapies, solidifying Orthogon’s leadership in addressing barriers to polyomavirus treatments.
The company’s therapeutic strategy offers the first comprehensive solution to managing the full BK virus risk ladder—from early reactivation, indicated by virus shedding in the urine, to systemic spread and severe complications such as BKVAN, graft dysfunction, and transplant failure. By intervening at the earliest stages of reactivation, this approach extends treatment benefits to more patients, reduces the need for aggressive late-stage interventions, and improves long-term transplant success.
“It is incredibly rewarding to see our drug designs progress from the bench into translational models,” said Ali H. Munawar, Ph.D., CEO of Orthogon Therapeutics. “Our small molecule antivirals offer unique advantages over emerging therapies, overcoming challenges in delivery, stability, and—most importantly—reaching the intracellular sites of viral replication in the kidney. These capabilities, combined with the dosing flexibility of oral administration, enable us to close significant gaps in transplant patient care.”
This strategic raise underscores Orthogon’s disciplined approach to capitalization, enabling focused progress on high-impact milestones and reinforcing investor confidence in its mission to deliver the first orally administered therapy for BK polyomavirus infections.
About BK and Polyomaviruses:
BK virus (BKV), a member of the polyomavirus family, establishes a lifelong, persistent yet silent infection in over 80% of healthy adults. BK virus reactivation occurs in the kidneys of nearly half of all solid organ and stem cell transplant patients, leading to severe complications and graft loss. Other human polyomaviruses, such as JC virus and Merkel Cell polyomavirus, cause fatal progressive multifocal leukoencephalopathy (PML) and aggressive Merkel cell carcinoma.
About Orthogon Therapeutics:
Orthogon leverages advanced structure-based drug design and biophysics to create groundbreaking therapies for challenging drug targets. To learn more, visit www.orthogontherapeutics.com.
Orthogon Therapeutics, LLC is an independent, privately held R&D company affiliated with the Pledge Therapeutics discovery platform. The company is headquartered in the biotech hub of Greater Boston with a branch in Leuven, Belgium. More info at www.pledge-tx.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20241210996847/en/
Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
Following Oral Presentation of Phase I Data at AACR 2026, Debiopharm Announces FDA Fast Track Designation for Lunresertib in Combination With Zedoresertib for Genomic-Defined Platinum-Resistant Ovarian Cancer20.4.2026 14:30:00 CEST | Press release
Debiopharm (www.debiopharm.com), a privately-owned, Swiss-based biopharmaceutical company aiming to establish tomorrow’s standard of care to cure cancer and infectious diseases, today announced that the U.S. Food and Drug Administration (FDA) has granted Fast Track designation to the combination of its PKMYT1 inhibitor, lunresertib (Debio2513), and its WEE1 inhibitor, zedoresertib (Debio 0123). The designation is for the treatment of adult patients with CCNE1 amplified, or a deleterious mutation in either FBXW7 or PPP2R1A, platinum-resistant/refractory ovarian cancer. The FDA’s Fast Track program is designed to facilitate the development and expedite the review of new drugs intended to treat serious conditions and fill an unmet medical need. Programs granted Fast Track designation benefit from more frequent communication with the FDA and, if relevant criteria are met, may be eligible for Priority Review and Accelerated Approval of a New Drug Application (NDA). Momentum Following AACR O
Data4Industry-X Empowers Industrial Organizations in Meeting Digital Product Passport Requirements20.4.2026 14:17:00 CEST | Press release
Contributing to the International Manufacturing-X Council Showcase at Hannover Messe, to improve resilience, productivity and innovation in Manufacturing Hannover Messe - Data4Industry-X, the trusted industry data space solution, builds its momentum by accelerating Digital Product Passport (DPP) compliance with trusted, secure and traceable data exchange at scale, as demonstrated at Hannover Messe April 20-24, 2026 . As theDigital Product Passport becomes a regulatory reality for manufacturing organizations, the ability to exchange data in a trusted, secure and traceable environment across the entire supply chain, in compliance with data regulations such as the Data Act, has become critical. Actively contributing to the International Manufacturing-X Council showcase, driven by LNI 4.0 association involving 16 countries, Data4Industry-X, a decentralized environment, demonstrates the use case of the DPP on the battery’s State of Health, and how the battery current capacity and performanc
Ushio Industry & Entertainment Appoints Takuya Matsumoto as President & CEO and Outlines Strategic Direction for the Next Phase20.4.2026 12:32:00 CEST | Press release
Ushio Industry & Entertainment (Ushio INE GmbH) announces the appointment of Takuya Matsumoto as President and Chief Executive Officer, marking a key milestone in the evolution of the business and its integration within the Ushio Group. With more than 20 years of experience across sales, new business development, and global operations, Takuya Matsumoto brings deep industry expertise and a strong leadership track record within Ushio. Most recently, he served as General Manager of the Global Business Unit, where he played a pivotal role in advancing global growth initiatives. A Clear Vision: A strong heritage with a forward-looking vision focused on innovation, operational excellence, and customer value Under Takuya Matsumoto’s leadership, Ushio INE will continue to build on its strong legacy while advancing its mission to deliver high-performance, high-quality lighting solutions across industrial and entertainment markets worldwide. “Our mission is to continuously deliver essential ligh
Compass Pathways to Participate in Needham Virtual Psychedelics Forum on April 27, 202620.4.2026 12:30:00 CEST | Press release
Compass Pathways plc (Nasdaq: CMPS), a biotechnology company dedicated to accelerating patient access to evidence-based innovation in mental health, announced today that CEO Kabir Nath will participate in a panel entitled “Clinical Risk, Regulatory Reality and the Path to Scale” at the Needham Virtual Psychedelics Forum on April 27, 2026, at 11am-12pm ET. A live audio webcast of this event will be accessible from the “Events” page of the Investors section of the Compass website. A replay of the webcast will be accessible for 30 days following each event. About Compass Pathways Compass Pathways plc (Nasdaq: CMPS) is a biotechnology company dedicated to accelerating patient access to evidence-based innovation in mental health. We are motivated by the need to find better ways to help and empower people with serious mental health conditions who are not helped by existing treatments. We are pioneering a new paradigm for treating mental health conditions focused on rapid and durable response
PPG invests in testing line for radiation-curable coatings at Marly, France20.4.2026 11:00:00 CEST | Press release
Installation helps accelerate development cycles, reduce number of customer trials PPG (NYSE: PPG) today announced the installation of an advanced testing line for radiation-curable coatings at its R&D Center of Excellence in Marly, France. The line can test multiple curing technologies, including infrared (IR), ultraviolet (LED, excimer and arc lamps), and electron beam (EB). This investment allows the company to accurately replicate customer production conditions, helping accelerate development cycles and reduce the number of customer trials. Unlike conventional thermal curing, radiation curing requires less energy because it operates at or near ambient temperature. This reduction in energy demand can significantly lower carbon emissions by 65%*, with further gains possible when powered by renewable energy sources. In addition, UV- and EB-curable systems typically use 100% solids formulations with no solvents, reducing or eliminating volatile organic compound emissions. Beyond sustai
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom