Kioxia Develops OCTRAM (Oxide-Semiconductor Channel Transistor DRAM) Technology
10.12.2024 09:22:00 CET | Business Wire | Press release
Kioxia Corporation, a world leader in memory solutions, today announced the development of OCTRAM (Oxide-Semiconductor Channel Transistor DRAM), a new type of 4F2 DRAM, comprised of an oxide-semiconductor transistor that has a high ON current, and an ultra-low OFF current, simultaneously. This technology is expected to realize a low power DRAM by bringing out the ultra-low leakage property of the InGaZnO*1 transistor. This was first announced at the IEEE International Electron Devices Meeting (IEDM) held in San Francisco, CA on December 9, 2024. This achievement was jointly developed by Nanya Technology and Kioxia Corporation. This technology has the potential to lower power consumption in a wide range of applications, including AI and post-5G communication systems, and IoT products.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20241209099702/en/
Fig.1: Cross-sectional TEM image for the InGaZnO vertical transistor (Photo: Business Wire)
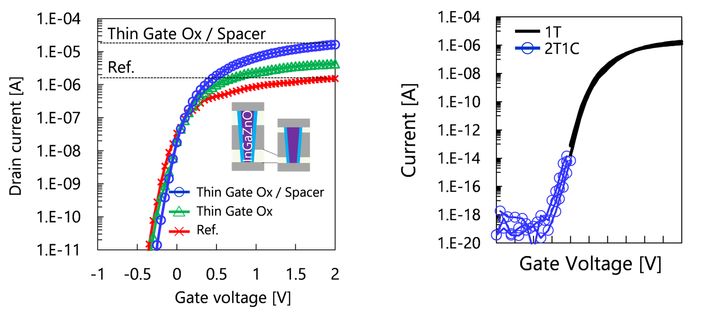
The OCTRAM utilizes a cylinder-shaped InGaZnO vertical transistor (Fig.1) as a cell transistor. This design enables the adaptation of a 4F2 DRAM, which offers significant advantages in memory density compared to the conventional silicon-based 6F2 DRAM.
The InGaZnO vertical transistor achieves a high ON current of over 15μA/cell (1.5 x 10-5 A/cell) and an ultra-low OFF current below 1aA/cell (1.0 x 10-18 A/cell) through device and process optimization (Fig.2). In the OCTRAM structure, the InGaZnO vertical transistor is integrated on top of a high aspect ratio capacitor (capacitor-first process). This arrangement allows for the decoupling of the interaction between the advanced capacitor process and the InGaZnO performance (Fig.3).
*1: InGaZnO is a compound of In(indium), Ga(gallium), Zn(zinc), and O(oxygen)
- This announcement has been prepared to provide information on our business and does not constitute or form part of an offer or invitation to sell or a solicitation of an offer to buy or subscribe for or otherwise acquire any securities in any jurisdiction or an inducement to engage in investment activity nor shall it form the basis of or be relied on in connection with any contract thereof.
- Information in this document, including product prices and specifications, content of services and contact information, is correct on the date of the announcement but is subject to change without prior notice.
About Kioxia
Kioxia is a world leader in memory solutions, dedicated to the development, production and sale of flash memory and solid-state drives (SSDs). In April 2017, its predecessor Toshiba Memory was spun off from Toshiba Corporation, the company that invented NAND flash memory in 1987. Kioxia is committed to uplifting the world with memory by offering products, services and systems that create choice for customers and memory-based value for society. Kioxia's innovative 3D flash memory technology, BiCS FLASH™, is shaping the future of storage in high-density applications, including advanced smartphones, PCs, SSDs, automotive and data centers.
View source version on businesswire.com: https://www.businesswire.com/news/home/20241209099702/en/

Subscribe to releases from Business Wire
Subscribe to all the latest releases from Business Wire by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from Business Wire
NIQ (NYSE: NIQ) Unveils Six New AI-Powered Capabilities at C360, Executing on Its Vision for Agentic Commerce9.6.2026 21:00:00 CEST | Press release
New capabilities help organizations operationalize trusted market intelligence across enterprise AI environments and workflows, while preparing for the future of agentic commerce NielsenIQ (NYSE: NIQ) today unveiled six new AI-powered capabilities at C360, the company's flagship client and industry event, marking a major milestone in the execution of NIQ’s AI strategy. Thenew capabilities help brands, retailers, and other organizations operationalize trusted market intelligence across enterprise workflows, AI environments, and the emerging world of agentic commerce. NIQ’s AI value creation strategy is built on two foundational principles: Delivering The Full View™: Combining the industry’s broadest and deepest datasets, advanced AI models, and rich content to help organizations unlock actionable insights and drive AI innovation at scale. Enabling AI-Powered Capabilities: Delivering trusted intelligence and proprietary IP through NIQ’s advanced AI applications, assistants, and agents em
H55 Delivers Certification-Grade Propulsion Battery Modules to Pratt & Whitney Canada, Supporting Demonstration of Hybrid-Electric Aircraft Technology9.6.2026 17:31:00 CEST | Press release
Production-conforming systems manufactured in a regulator-approved facility mark H55’s transition from technology development to commercialization of certification-grade energy storage solutions for hybrid-electric aviation. H55 today announced the delivery of certification-grade propulsion battery modules to Pratt & Whitney Canada in support of the RTX Hybrid-Electric Flight Demonstrator — a milestone that further validates H55’s transition from technology development to industrial-scale execution and represents an important step in the commercialization of the company’s certification-grade energy storage technologies for hybrid-electric aerospace applications. Pratt & Whitney is an RTX business. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609825006/en/ H55 Adagio Battery Modules in Pratt & Whitney Canada Hangar ©RTX The delivery represents more than hardware integration. It demonstrates H55’s ability to manufacture p
New Polymorph of Indomethacin Discovered9.6.2026 17:00:00 CEST | Press release
- Joint Research Involving Rigaku Published in Crystal Growth & Design - Rigaku Corporation, a global solution partner in X-ray analytical systems and a group company of Rigaku Holdings Corporation (headquarters: Akishima, Tokyo; CEO: Jun Kawakami; “Rigaku”), announced that the results of a joint research project conducted with Shionogi & Co., Ltd., JEOL Ltd., and Meiji Pharmaceutical University have been published in Crystal Growth & Design, a world-renowned international journal in the field of crystallography. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260609347405/en/ Cover of the June 2026 issue of Crystal Growth & Design, featuring this research. This research uncovered a previously unknown polymorph (κ-form) of indomethacin, a widely used pain relief and anti-inflammatory drug. The research team also conducted structural analysis and characterization of the newly identified crystal form. Although indomethacin has
SLB Collaborates with Qualcomm on Edge AI Solutions for Energy Operations9.6.2026 16:28:00 CEST | Press release
Collaboration combines edge computing and energy workflows to support real-time operational decision-making Global energy technology company SLB (NYSE: SLB) today announced a memorandum of understanding with Qualcomm Technologies, Inc. to enable edge AI solutions for the energy industry, supporting real-time operational decision-making across wells, facilities and production systems. The collaboration combines Qualcomm Technologies’ low-power edge computing and AI processing capabilities, with SLB’s Agora™ edge AI and IoT solutions developed for remote and operationally complex environments. “Together, SLB and Qualcomm Technologies aim to help operators apply AI more effectively across energy infrastructure,” said Rakesh Jaggi, president, Digital, SLB. “Many energy operations rely on real-time decision-making in remote environments where connectivity and responsiveness directly affect performance. AI systems designed around the realities of energy operations can help support more consi
RevBits and Stony Brook University’s Ethos Lab Establish a Collaborative Partnership to Further the Field of Cyber Security Education and Application9.6.2026 16:15:00 CEST | Press release
Through the partnership, RevBits will provide its full suite of cybersecurity solutions to Stony Brook University’s Ethos Lab, to deliver the capability to advance student education and equip them with the knowledge needed to face modern cyber threats in a realistic, contained environment. Stony Brook University’s (SBU) Ethos Lab, in the College of Engineering and Applied Sciences Department of Computer Science, recently announced a collaborative partnership with RevBits, LLC. Through the partnership, SBU’s Ethos Lab will utilize the RevBits solutions suite to build computer science labs and a cybersecurity-focused curriculum, reinforcing lessons from the classroom. The platform simulates environments containing five major threat landscapes, including Endpoint Security, Privileged Access Management, Email Security, Zero Trust Network and Deception Technology. SBU’s Department of Computer Science was recently designated as a National Center of Academic Excellence in Cybersecurity Resear
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom